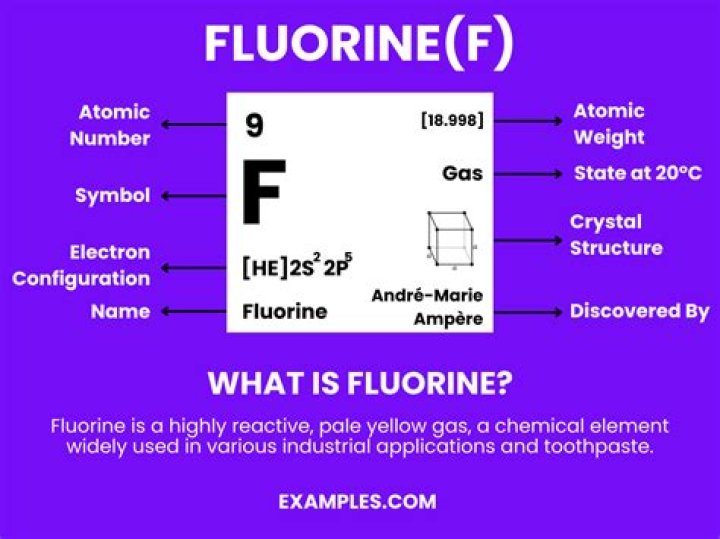
Anomalous Behaviour of Fluorine Fluorine differs from rest of the members of its group because of its small size, high electro negativity and non availability of d orbitals in the valence shell..
Besides, what is the anomalous Behaviour of fluorine?
Fluorine is anomalous in many properties like, ionisation enthalpy, electronegativity, enthalpy of bond dissociation that are higher than expected from the regular trends among the halogens. Its ionic and covalent radii, melting and boiling points, and electron gain enthalpy is quite lower than expected.
Similarly, what do you mean by anomalous Behaviour? Anomalous means differing from the norm for a given situation, psychologically speaking that is. Anomalous behavior would be behavior that is unusual for the circumstances. Of course there are many different degrees of what is considered to be normal and anomalous behavior.
Just so, why does Oxygen show anomalous Behaviour in its group?
Oxygen shows anomalous behaviour due to formation of p(pi)-p(pi) bond. Oxygen has -2 oxidation state ,while all the other elements have +4 &+6 oxidation state.
How does fluorine differ from other halogens?
The main points of difference are : (1) Fluorine is most reactive of all the halogens due to lower value of F — F bond dissociation energy (F2 = 158, Cl2 = 243, bromine = 192 and iodine = 151 kJ mol–1) . Other halogens show positive oxidation states of +1, +3, +5 and +7.
Related Question Answers
What is anomalous lithium behavior?
The anomalous behaviour of lithium is as follows: Lithium is harder than other metals of the respective group. It has higher Melting and boiling point than other metals. It is least reactive. As the polarizing power decreases with the increasing size of the ions the other alkali metal ions usually form anhydrous salts.Why fluorine does not form Polyhalides?
Fluorine, because of the absence of d orbitals in its valency shell, does not formany polyhalides. Other halogens frequently associate with the halide ions forming polyhalide ions because of the vacant d orbitals in their valency shell.Why does fluorine differ from other members of its group?
Fluorine differs from rest of the members of its group because of its small size, high electro negativity and non availability of d orbitals in the valence shell. The oxidation state of F in its compound is always –1 while other elements exhibit a large number of oxidation states (–1, +1, +3, +5, +6, +7).What is peculiar character of fluorine?
Characteristics: Fluorine is the most reactive and the most electronegative of all the elements. Fluorine is a pale yellow, diatomic, highly corrosive, flammable gas, with a pungent odor. It is the lightest halogen. It reacts violently with water to produce oxygen and the extremely corrosive hydrofluoric acid.What properties are responsible for the anomalous Behaviour of carbon?
The Anomalous Behaviour of Carbon We can attribute this behaviour to carbon mainly due to : The small size of the atom. High electronegativity. High ionization enthalpy.Is of2 an oxide of fluorine?
OF2 is known as difluoride and not fluorine oxide .What are the anomalous properties of water?
Anomalous water, also called Orthowater, orPolywater, liquid water generally formed by condensation of water vapour in tiny glass or fused-quartz capillaries and with properties very different from those well established for ordinary water; e.g., lower vapour pressure, lower freezing temperature, higher density andWhy does boron show anomalous?
Boron ,the first member of group 13 elements, shows anomalous behaviour and differ from rest of the members of its family. The main reason for this difference are : 1)Boron being small is harder than the other elements of its group. 2)It has higher melting and boiling point than those of the other members of its group.What is anomalous Behaviour of oxygen?
Anomalous behaviour of oxygen Oxygen forms strong hydrogen bonding in H2O which is not found in H2S. Also, the maximum covalency of oxygen is four, whereas in a case of other elements of the group, the valence shells can be expanded and covalency exceeds four.What is nascent oxygen?
The term nascent oxygen refers to oxygen that has just been made, presumably in a chemical reaction. Some people interpret this to mean O2 , which is made in many reactions, while others believe it to be a monatomic oxygen radical, which is far more reactive.What are the anomalous properties of nitrogen?
Anomalous behaviour of nitrogen: I)Nitrogen differ from of the members of this group due to its small size, high electronegativity, high ionisation enthalpy and non-availability of d orbitals. pπ -pπ multiple bonds with itself and with other elements having small size and high electronegativity (e.g., C, O).Why does the first element in each group of P Block show anomalous Behaviour?
The anomalous behaviour of first element of s and p block elements of each group as compared to other group members is due to following reasons: Small size of atom, large charge/radius ratio, high electronegativity and non availability of d-orbitals in their valence shell.How do you study anomalous behavior?
There are five steps to study the anomalous behavior of water by using Hope's apparatus. They are, Step 1: A beaker filled with water is cooled along with the Hope's device an hour before conducting the experiment. Step 2: A cooling mixture is prepared using crushed ice and kitchen salt.What is analogous Behaviour?
Brame, 1998), we used data from the Cambridge Study in Delinquent Development to investigate the association between criminal activity and a set of so-called “analogous behaviors” (i.e., excessive drinking, smoking, gambling, involvement in accidents, etc.).What is the importance anomalous Behaviour of water?
The anomalous expansion of water refers to the fact that water, unlike most substances, expands when it freezes. The density of water increases as temperature decreases, but reaches a maximum at 4 degrees C then begins to expand. The significance of this is that ice is less dense than water so it floats in water.What is the meaning of anomalous verb?
The term "anomalous verb" is defined in A.S.Hornby'sA Guide to Patterns and Usage in English. Anomalous verbs are verbs that do not take "to do" in questions or negative sentences (can, may, be, will, shall . . . ). It is a useful notion, because it is not exactly the same as "auxiliary verb". (What is the anomalous Behaviour of lithium?
Anomalous behaviour of Lithium is due to 1) very small size of lithium atom and its ions. 2) higher polarising power of Li+ resulting in increased covalent character of its compounds which is responsible for their solubility in organic solvents.What is anomalous Behaviour of beryllium?
Anomalous Behaviour of Beryllium Beryllium do not impart color during flame test. Beryllium is small in size with high ionization enthalpy compared to other alkaline earth metals. Beryllium do not form oxides similar to other alkaline earth metals.What is anomalous properties of second period elements?
Anomalous Periodic Properties of Second Period Elements. It has been observed that Lithium, Beryllium, Boron, Carbon, Nitrogen, Oxygen, and Fluorine have slightly different periodic properties than the rest of the elements belonging to Groups 1, 2, 13-17 respectively.