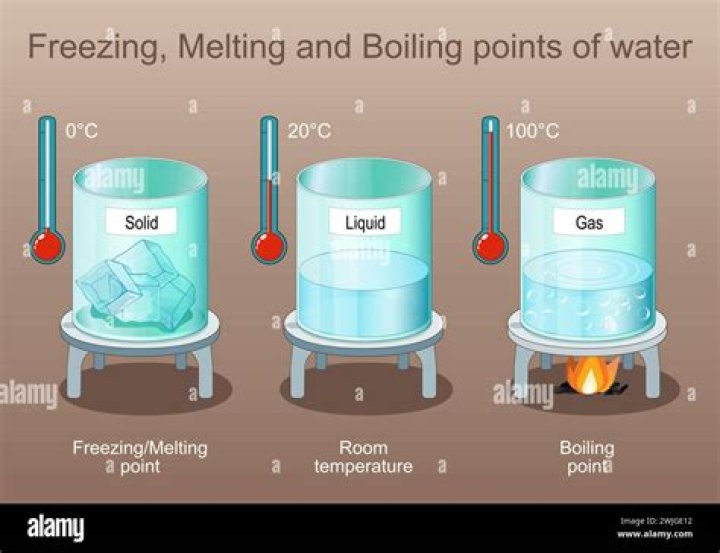
So, when liquid is boiled then its molecules spread awayand turns into gas. That's called Evaporation. But whensolid is heated (like ice, iron or such matels etc.) Simply,transformation of liquid to gas is called Evaporation andtransformation of solid to liquid is calledMelting..
In respect to this, what is the difference between melting and evaporation?
As nouns the difference between vaporization andmelting is that vaporization is a conversion of a solidor a liquid into a gas while melting is the process ofchanging the state of a substance from solid to liquid by heatingit past its melting point.
Beside above, what are some examples of melting? Examples include:
- Melting Ice to liquid water.
- Melting of steel (requires very high temperature)
- Melting of mercury and Gallium (both are liquid at roomtemperature)
- Melting of butter.
- Melting of candle.
Secondly, what is the difference between melting and boiling?
The main difference between boiling point andmelting point is that the melting point is defined asthe temperature at which solid and liquid phases are inequilibrium, whereas the boiling point is the temperature atwhich the vapour pressure of a liquid is equal to the externalpressure.
What happens to particles when they melt?
When a solid is heated the particles gain energyand start to vibrate faster and faster. The particles in theliquid are the same as in the solid but they have moreenergy. To melt a solid energy is required to overcome theattractions between the particles and allow them to pullthem apart.
Related Question Answers
What are the 6 changes of state?
There are six changes of phase that substances gothrough: - Freezing: liquid to solid.
- Melting: solid to liquid.
- Condensation: gas to liquid.
- Vaporization: liquid to gas.
- Sublimation: solid to gas.
- Deposition: gas to solid.
What happens during a phase change?
They are changes in bonding energy between themolecules. If heat is coming into a substance during a phasechange, then this energy is used to break the bonds between themolecules of the substance. The heat is used to break the bondsbetween the ice molecules as they turn into a liquidphase.What are 3 examples of deposition?
One example of deposition is the process bywhich, in sub-freezing air, water vapor changes directly to icewithout first becoming a liquid. This is how frost and hoar frostform on the ground or other surfaces. Another example iswhen frost forms on a leaf.Is sublimation endothermic or exothermic?
Fusion, vaporization, and sublimation areendothermic processes, whereas freezing, condensation, anddeposition are exothermic processes. Changes of state areexamples of phase changes, or phase transitions. All phase changesare accompanied by changes in the energy of a system.What is a phase change in physics?
Phase changes: it takes energy to changesphases from a solid to a liquid and from a liquid to a gas. Thesubstance releases energy when changing phase from gas toliquid or from liquid to solid. During a phase change, thenumber of degrees of freedom changes, and so does thespecific heat capacity.What is sublimation and deposition?
Deposition occurs when a substance goes directlyfrom the gas state to the solid state. Like sublimation, theintermediate liquid phase is skipped over. In contrast tosublimation, the process of deposition releasesenergy. An example of deposition is the formation offrost.What are the changes in matter?
Changes of state are physical changes inmatter. They are reversible changes that do not involvechanges in matter's chemical makeup or chemical properties.Common changes of state include melting, freezing,sublimation, deposition, condensation, andvaporization.What is boiling point in physics?
The boiling point of a substance is thetemperature at which the vapor pressure of a liquid equalsthe pressure surrounding the liquid and the liquid changes into avapor. A liquid at high pressure has a higher boiling pointthan when that liquid is at atmospheric pressure.What increases melting point?
The force of attraction between the molecules andsymmetry affects the melting point of a compound. Strongerintermolecular interactions result in higher melting points.Ionic compounds usually have high melting points because theelectrostatic forces holding the ions (ion-ion interaction) aremuch stronger.Does higher boiling point mean higher melting point?
Higher melting and boiling points signifystronger noncovalent intermolecular forces. As you would expect,the strength of intermolecular hydrogen bonding and dipole-dipoleinteractions is reflected in higher boilingpoints.Is Melting Point a physical property?
Physical properties can be observed or measuredwithout changing the composition of matter. Physicalproperties are used to observe and describe matter. Physicalproperties include: appearance, texture, color, odor,melting point, boiling point, density, solubility,polarity, and many others.What is high melting and boiling point?
Melting point and boiling point. When ametal melts or boils, this is a change of physical state. Thisenergy is needed to overcome the forces of attraction between themetal ions and the delocalised electrons in the metal. The moreenergy needed, the higher the melting point orboiling point .What is melting point of a liquid?
The melting point (or, rarely, liquefactionpoint) of a substance is the temperature at which itchanges state from solid to liquid. At the meltingpoint the solid and liquid phase exist inequilibrium.What is melting and melting point?
Melting is a process in which a solid substancechanges its state into a liquid. melting point is apoint at which a particular solid substance startsmelting. melting point denotes the temperature atwhich is solid state starts getting converted into a liquid statein a process called melting. 4.2. 10 votes.Does everything have a melting point?
No in the sense that a material must maintain anidentical chemical makeup and then change from one phase toanother, not all solids have a melting point. Changing froma solid to a liquid state such as when a metal, wax or ice isheated to its melting point, or the specific temperature atwhich melting occurs.What is the process of melting?
Melting is a process that causes asubstance to change from a solid to a liquid. Melting occurswhen the molecules of a solid speed up enough that the motionovercomes the attractions so that the molecules can move past eachother as a liquid.How do you explain melting?
Melting, or fusion, is a physical process thatresults in the phase transition of a substance from a solid to aliquid. This occurs when the internal energy of the solidincreases, typically by the application of heat or pressure, whichincreases the substance's temperature to the meltingpoint.What is melting short answer?
Melting is the change of state from (crystalline)solid to liquid. It occurs at temperatures above the meltingpoint, which is the temperature at which solid and liquid can existat equilibrium. Ice melts to liquid water at temperaturesabove 0 °C.What does melting point depend on?
A strong attraction between molecules results in ahigher melting point. In organic compounds, the presence ofpolarity, especially hydrogen bonding, usually leads to a highermelting point. The melting points of polar substancesare higher than the melting points of nonpolar substanceswith similar sizes.