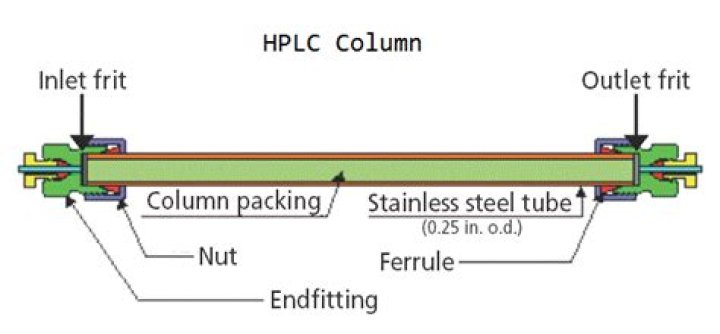
Why Silica is used in HPLC column?
.
Herein, why c18 column is used in HPLC?
C18 columns are HPLC (high performance liquid chromatography) columns that use a C18 substance as the stationary phase. C18 HPLC columns are used in environmental sciences and chemical analysis, as well as industries such as pharmaceutical and environmental sciences, to analyze individual parts of chemical mixtures.
Furthermore, what are HPLC columns made of? HPLC columns are usually packed with pellicular, or porous particles. Pellicular particles are made from polymer, or glass beads. Pellicular particles are surrounded by a thin uniform layer of silica, polystyrene-divinyl-benzene synthetic resin, alumina, or other type of ion-exchange resin.
Secondly, why is silica used in chromatography?
Silica gel is a polar adsorbent and being slightly acidic in nature, it has a powerful capacity to absorb basic contents that may be present in the material that needs separation or purification. It is also well known for its role in reversed-phase partition chromatography.
Which silica is used for column chromatography?
The two most common stationary phases for column chromatography are silica gel (SiO2) and alumina (Al2O3), with the most commonly used mobile phases being organic solvents.
Related Question AnswersIs c18 polar or nonpolar?
High performance liquid chromatography (HPLC) stationary phases can be segregated by their ability to separate either polar on nonpolar compounds, that is, reversed-phase materials (C18, C8) strongly retain nonpolar solutes with polar solutes eluting at or near the void volume, and hydrophilic interactionWhat is ODS and BDS column?
columns. Both involve octadecasilane chain. Finally I came to know that ods contains free -OH functional group while bds i.e. base deactivated silica column has -OH groups deactivated or blocked. This is the reason why bds columns are also called as endcap columns.Why acetonitrile is used in HPLC?
The B solvent is generally an HPLC grade organic solvent such as acetonitrile or methanol with 0.1% acid. The acid is used to the improve the chromatographic peak shape and to provide a source of protons in reverse phase LC/MS. In our work we use acetonitrile as our organic solvent.What is difference between c8 and c18 column?
The key difference between C8 and C18 column is that the C8 column has Octylsilane as the stationary phase whereas the C18 column has Octadecylsilane. The C8 and C18 columns differ from each other according to the stationary phase. We use these columns in HPLC (high-performance liquid chromatography).Which is more polar c8 or c18?
C18 has a longer carbon chain, but C8 has a shorter one. C18 has higher retention while C8 has shorter retention. C18 has higher hydrophobicity, but C8 has a lower hydrophobicity.What is l7 column?
Merck offers a broad range of HPLC columns that are fully compliant with USP (United States Pharmacopoeia) specifications. L7 USP columns are described as octyl silane (C8), chemically linked to porous silica particles, 1.5 to 10 µm in diameter.Is acetonitrile polar?
Acetonitrile has a 5.8 polarity index. Polarity just means opposite charges in certain parts of a molecule, even if the overall charge is neutral (i. e. it's not a positive or negative ion). Water (10.2 index) is polar and most oils are non-polar (e.g. Petroleum ether = 0.1 index).Why phosphate buffer is used in HPLC?
What are the common buffers used in HPLC? Since the retention of ionizable compounds is very sensitive to the mobile phase pH, it is necessary to control the pH of the mobile phase by the addition of a buffer. Phosphate and acetate are particularly useful buffers because they can be used at wavelengths below 220 nm.What is Rf value?
The Rf value is defined as the ratio of the distance moved by the solute (i.e. the dye or pigment under test) and the distance moved by the the solvent (known as the Solvent front) along the paper, where both distances are measured from the common Origin or Application Baseline, that is the point where the sample isIs silica polar or nonpolar?
silica gel is very polar. so more polar material moves more slowly than nonpolar material, which feels less attraction from the silica gel. it's used in TLC and column chromatography (not paper chromatography).What is the formula of silica gel?
Silica gel, chemical formula: mSiO2 • nH2O, appears as slightly transparent white solid substance. It belongs to genus amorphous. Silica is a highly active, porous material with the internal surface area being large. The specific surface area per g silica can reach above 450 m2.Is silica gel acidic or basic?
Silica and alumina are both polar adsorbents so the more polar components in the mixture to be separated are retained more strongly on the stationary phase and are therefore eluted from the column last. Silica is recommended for most compounds, but as it is slightly acidic, it preferentially retains basic compounds.Why silica gel is used in bags?
Silica gel packets are used to absorb moisture and keep things dry. Many manufacturers add these desiccant packs to their new products to keep them fresh and moisture-free for the consumer.Why silica gel is used in TLC?
Silica gel is by far the most widely used adsorbent and remains the dominant stationary phase for TLC. The surface of silica gel with the highest concentration of geminal and associated silanols is favored most for the chromatography of basic compounds because these silanols are less acidic.Why is silica gel used in bags?
Silica gel packets are used to absorb moisture and keep things dry. Many manufacturers add these desiccant packs to their new products to keep them fresh and moisture-free for the consumer.What is the chemical name of silica gel?
Silica gel is an amorphous substance. Its chemical formula is mSiO2. nH2O. It is insoluble in water and any solvent, non-toxic, odorless, stable in chemical properties, and does not react with any substances except strong alkali and hydrofluoric acid.How do you pack silica gel columns?
Packing a (silica gel) column:- Use a piece of wire to add a plug of cotton to the bottom of the column.
- Clamp the column to a ring stand and add enough sand to fill the curved portion of the column.
- Place a pinch clamp on the tubing, then fill the column 1/4 to 1/3 full with the intial eluent.