Why do different solutions of silver nanoparticles all composed of the same material have different colors?
Why do different solutions of silver nanoparticles all composed of the same material have different colors?
Nanoparticles possess wondrous optical, electronic, magnetic, and catalytic properties than the bulk material owing to their high surface area to volume ratio [4, 5]. Metal nanoparticles like silver and gold show different colors due to their Surface Plasmon Resonance (SPR) phenomenon.
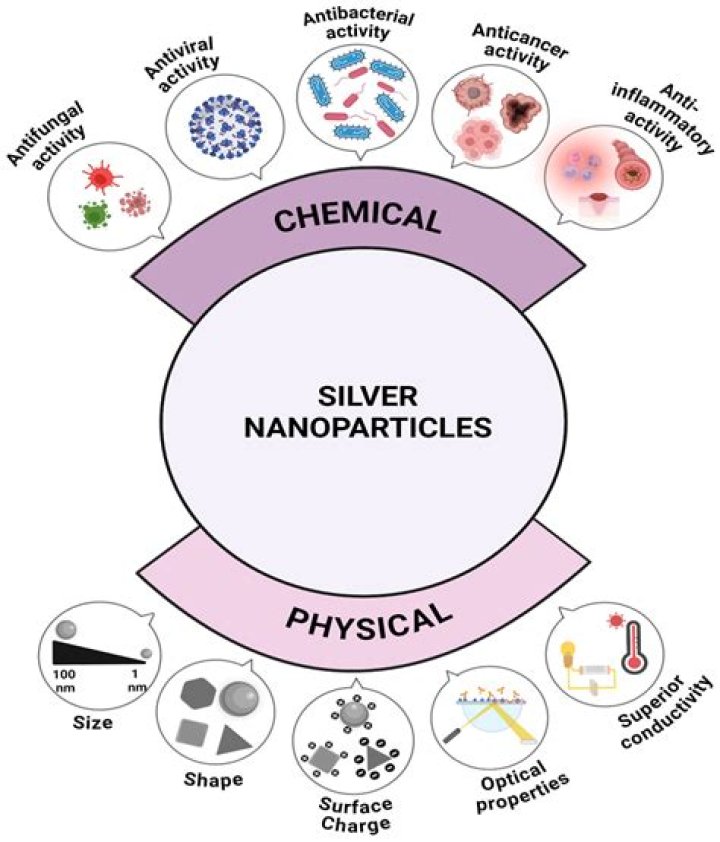
What are the benefits of silver nanoparticles?
The antibacterial effects of silver nanoparticles have been used to control bacterial growth in a variety of applications, including dental work, surgery applications, wounds and burns treatment, and biomedical devices. It is well known that silver ions and silverbased compounds are highly toxic to microorganisms.
What are silver nanoparticles?
Silver nanoparticles are nanoparticles of silver of between 1 nm and 100 nm in size. Commonly used silver nanoparticles are spherical, but diamond, octagonal, and thin sheets are also common. Their extremely large surface area permits the coordination of a vast number of ligands.
How do you dissolve silver nanoparticles?
Protocol I used:
- culture of endophytes in 250 mL flask using LB media for 48 hrs at 37 deg C,
- centrifuge at 10,000 rpm for 10 min,
- AgNO3 solution (1 mM) added into 100 mL filtrate (1:1, v/v),
- addition of PVP (5 mL, 1% w/v),
- Incubation at room temperature in an incubator shaker (180 rpm).
Why silver nanoparticles are yellow?
This color change is due to the excitation of the surface plasmon resonance (SPR), as previously reported [27] [28][29]. The Spondia mombins seed extract was used as a reduction and capping molecules, as shown in Fig. 1, leading to the formation of silver nanoparticles. …
Why do silver nanoparticles appear yellow?
Size distribution (nm) If the stirring is continued once all of silver nitrate has been added, can cause aggregation that resulted yellow colloidal first and turn a dark yellow, then violet and eventually grey. Aggregation of silver nanoparticles also can cause the colloidal breaks down and particle settle out.
Are silver nanoparticles harmful to humans?
The researchers found that silver nanoparticles had a toxic effect on cells, suppressing cellular growth and multiplication and causing cell death depending on concentrations and duration of exposure. In particular, the 200 nm silver particles caused a concentration-dependent increase in DNA damage in the human cells.
Are silver nanoparticles harmful?
How can you tell silver nanoparticles?
UV–Vis spectroscopy is the most important technique and the simplest way to confirm the formation of nanoparticles. The absorbance spectrum of the colloidal sample was obtained in the range of 200–800 nm, using a UV–Vis spectrometer Shimadzu-UV 1800 with distilled water as a reference.
Is Nano Silver soluble in water?
If the surface of a silver nanoparticle is passivated by cysteine, the dissolution is quantitatively inhibited. Upon immersion of silver nanoparticles in pure water for 8 months, leading to about 50% dissolution, no change in the surface was observed by transmission electron microscopy.
How do you dissolve nanoparticles?
If you want to dissolve the NPs (ie destroy them) you can use a strong acid, but if you want to diperse them ie make them stable in a colloidal suspension in a medium such as water you will have to add a surfactant that is a molecule wich will change the surface properties of you nanoparticles.