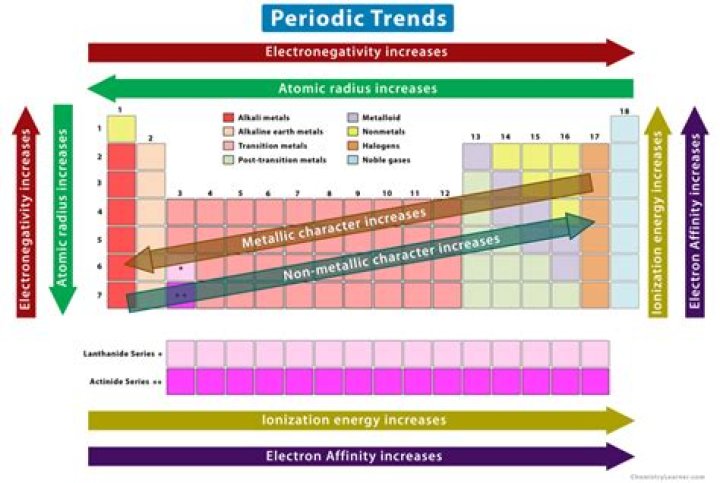
Major periodic trends include: electronegativity, ionization energy, electron affinity, atomic radius, melting point, and metallic character. These trends exist because of the similar atomic structure of the elements within their respective group families or periods, and because of the periodic nature of the elements..
Similarly, why is it still beneficial for chemists to understand periodic trends?
It helps them identify elements quicker. Barium, Iron, Bromine; Electronegativity decreases down a column and increases across a period.
Also, what properties follow a periodic trend? Periodic trends are specific patterns in the properties of chemical elements that are revealed in the periodic table of elements. Major periodic trends include electronegativity, ionization energy, electron affinity, atomic radii, ionic radius, metallic character, and chemical reactivity.
Thereof, what is the purpose of the periodic table and why is it important?
To summarize, the periodic table is important because it is organized to provide a great deal of information about elements and how they relate to one another in one easy-to-use reference. The table can be used to predict the properties of elements, even those that have not yet been discovered.
What is the trend in ionization energy across a period?
Ionization energy exhibits periodicity on the periodic table. The general trend is for ionization energy to increase moving from left to right across an element period. Moving left to right across a period, atomic radius decreases, so electrons are more attracted to the (closer) nucleus.
Related Question Answers
What is the trend in electronegativity?
The higher the electronegativity of an atom, the greater its ability to attract shared electrons. The electronegativity of atoms increases as you move from left to right across a period in the periodic table. The electronegativity of atoms decreases as you move from top to bottom down a group in the periodic table.Can the properties of an element be predicted using a periodic table?
Periodic Trends Can the properties of an element be predicted using a periodic table? It includes information about atomic masses and element symbols, but it can also be used to make predic- tions about atomic size, electronegativity, ionization energies, bonding, solubility, and reactivity.Why do metals have low ionization energy?
Why do metals have a low ionization energy? Because the valence electrons are farther away from the positively charge nucleus, so the force of attraction is low.Why would an atom want to gain electrons?
So atoms or elements, when they exist as ions, they want to lose/gain or share electrons to attain a stable electronic configuration and achieve an octet/duplet valence shell structure resembling that of noble gases. Hence cations react to lose electrons, anions react to gain electrons.What is the periodic trend for metal reactivity in a group?
Reactivity Trends Metal reactivity increases down a group because as nuclear shielding increases and the nucleus' hold on the valence electron weakens, therefore it is easier to remove valence electrons.Why does electronegativity increase?
Electronegativity increases as you move across the periodic table from left to right. This occurs due to a greater charge on the nucleus, causing the electron bonding pairs to be very attracted to atoms placed further right on the periodic table. Fluorine is the most electronegative element.Is specific heat capacity a periodic trend?
Why do heavier elements have smaller specific heat values. In the periodic table the trend is that as the mass of each element increase's the specific heat tends to go down.How are elements named?
In keeping with tradition, elements are named after a mythological concept or character (including an astronomical object); a mineral, or similar substance; a place or geographical region; a property of the element; or a scientist.What things can you learn from the periodic chart of the elements?
Within each element square, information on the element's symbol, atomic number, atomic mass, electronegativity, electron configuration, and valence numbers can be found. At the bottom of the periodic table is a two row block of elements that contain the lanthanoids and actinides.What does a periodic table tell you?
What does the Periodic Table tell us about elements? The atomic number tells you how many protons are in the element, which tells you the number of electrons because they have to balance. The numbers above the periodic table are the group numbers.Why are the elements important?
No elements, no matter. Elements are also important because of how they are formed. By lighter, I mean elements that are lighter than iron and nickel on the periodic table. These elements get thrown out into the universe during stellar explosions called novae.Is it important to memorize the periodic table?
No, you don't need to memorize the periodic table, except for pulling freak memory stunts. But knowing a lot of the properties and how they vary over the periodic system will really help you make connections and will make you think chemistry a lot faster.What is the most important thing to know about the periodic table?
The periodic table is one of the most important and basic topics of chemistry. It talks about all the elements present in nature and their atomic masses etc. The elements in the periodic table are arranged in rows and columns. Wherein rows are called 'periods' and columns are called 'groups'.Why is it called the periodic table?
The periodic table got its name from the way the elements are arranged in rows which are called periods. The columns of the table are called groups, some of which have specific names, such as the noble gases and the halogens. Going down the periodic table, the number of atomic orbitals increases by one for each row.Why are elements important to life?
Major Elements The six most common elements in living things are carbon, hydrogen, oxygen, nitrogen, phosphorus, and sulfur. Atoms of these elements combine and form thousands of large molecules. These large molecules make up the structures of cells and carry out many processes essential to life.How has the periodic table changed the world?
Mendeleev's chemical grid brought order to chemical chaos – and its rarer elements will shape the planet's future. This is because the periodic table is far more than just a list of the elements we know. It's a way of categorising and sorting them.What are the 3 periodic trends?
Major periodic trends include: electronegativity, ionization energy, electron affinity, atomic radius, melting point, and metallic character.Is Melting Point A periodic property?
Melting points and boiling points show periodic properties. This means that they vary in a regular way or pattern depending on their position in the Periodic Table.What do you mean by periodic properties?
'Periodic' means 'regularly recurring'. The chemical and physical properties of the chemical elements have similarities that recur, showing up in related elements in a group (column) of the periodic table. Such elements occur a definite number of places apart—such as every 8th, or every 18th, atomic number.