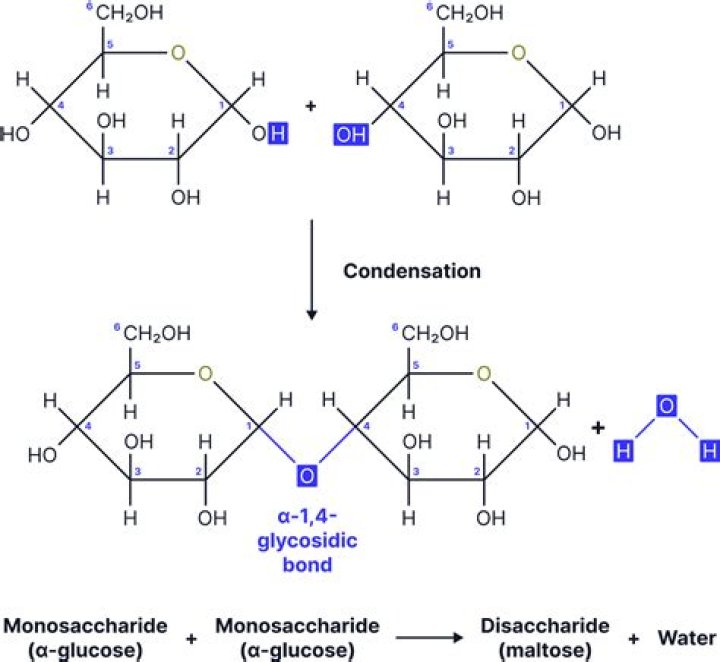
If heated to 200 °C, sucrose becomes caramel. When sucrose is hydrolyzed it forms an equimolar mixture of glucose and fructose. This mixture of monosaccharides is called invert sugar. Honeybees have enzymes called invertases that catalyze the hydrolysis of sucrose..
People also ask, what is formed when sucrose is hydrolyzed?
When sucrose is hydrolyzed it forms a 1:1 mixture of glucose and fructose. It is called invert sugar because the angle of the specific rotation of the plain polarized light changes from a positive to a negative value due to the presence of the optical isomers of the mixture of glucose and fructose sugars.
Also, how many monosaccharides are obtained by hydrolysis of sucrose? Sucrose, ordinary table sugar (cane or beet), is the pure organic chemical sold in greatest quantity. It is a disaccharide, which on hydrolysis gives one mole of glucose and one of fructose. The mixture of these two is called invert sugar, and is a major component of honey.
Also question is, what monosaccharides are produced from the hydrolysis of sucrose?
The hydrolysis of sucrose by boiling with a mineral acid, or by the enzyme invertase, produce a mixture of equal molecules of D-glucose and D-fructose.
What are the product of hydrolysis of cane sugar?
The product of the hydrolysis is a solution containing mainly sugars such as xylose, glucose and arabinose. Other compounds such as oligomers, furfural and acetic acid are also released.
Related Question Answers
What happens when sucrose is hydrolyzed?
When sucrose is hydrolyzed it forms a 1:1 mixture of glucose and fructose. It is called invert sugar because the angle of the specific rotation of the plain polarized light changes from a positive to a negative value due to the presence of the optical isomers of the mixture of glucose and fructose sugars.Why is sucrose bad for you?
Due to its simple chemical composition, sucrose is digested quickly. A serving of food rich in sucrose can cause a sharp increase in blood sugar that is often followed by a sharp decrease. The sudden rise and fall in blood sugar often affects mood, causing sudden bouts of irritability and fatigue.What does sucrose react with?
Sucrose melts and decomposes at 186 °C to form caramel, and when combusted produces carbon, carbon dioxide, and water. Reacting sucrose with sulfuric acid dehydrates the sucrose and forms elemental carbon, as demonstrated in the following equation: C12H22O11 + H2SO4 catalyst --->12 C + 11 H2O.What is the product of sucrose?
Sucrose is a disaccharide,commonly known as table sugar. Hydrolysis of sucrose yields equimolar mixture of D(+)glucose and D(-)fructose.What is the end product of sucrose?
In plants, glucose is derived from sucrose, which is the end product of photosynthesis or from storage carbohydrates. Sucrose is converted into glucose and fructose by the enzyme?What are the properties of sucrose?
Pure sucrose is most often prepared as a fine, colorless, odorless crystalline powder with a pleasing, sweet taste. Large crystals are sometimes precipitated from water solutions of sucrose onto a string (or other nucleation surface) to form rock candy, a confection.What happens when you heat sucrose?
What you are doing when you heat a sugar like sucrose is you are dehydrating it. The crystalline structure of sucrose breaks down and the molecules decompose into glucose and fructose and then lose water and then become isomers and polymerize to form caramel, a red-orange colored solid at room temperature.Is Sucrose a protein?
Fats are very simple architecturally and so are amino acids. Sucrose is a disaccharide of glucose bonded to fructose by a (beta 1,2-glycosidic linkage). This means bonds must break to utilize it making it a less efficient fuel source. If it were a protein, it'd be called sucrin.What is the process of hydrolysis?
In its simplest definition, hydrolysis is a chemical reaction in which water is used to break down the bonds of a particular substance. Hydrolysis can also be thought of as the exact opposite reaction to condensation, which is the process whereby two molecules combine to form one larger molecule.Why sucrose is called invertase?
Sucrase is also known as invertase or saccharase. It catalyzes the hydrolysis (breakdown) of sucrose (table sugar) to fructose and glucose, usually in the form of inverted sugar syrup. Invert sugar is the mixture of glucose and fructose produced by hydrolysis of sucrose, 1.3 times sweeter than sucrose.Is sucrose same as sugar?
Sugar. But, “SUGAR” refers only to sucrose, a disaccharide, made up of two sugars (glucose and fructose) bound together, that is naturally made by and found in all green plants. Sugar found in the food supply is harvested from sugar beets and sugar cane.Where is maltose found?
Maltose (or malt sugar) is an intermediate in the intestinal digestion (i.e., hydrolysis) of glycogen and starch, and is found in germinating grains (and other plants and vegetables). It consists of two molecules of glucose in an α-(1,4) glycosidic linkage.Is sucrose Dextrorotatory or Levorotatory?
Sucrose is dextrorotatory, but the resulting mixture of glucose and fructose is slightly levorotatory, because the levorotatory fructose has a greater molar rotation than the dextrorotatory glucose.What is galactose made of?
Medical Definition of Galactose Galactose: A sugar found in milk. Galactose is a disaccharide that is made up of two sugars, galactose and glucose, that are bound together.What is the product of starch hydrolysis?
Whenever starch (polysaccharides) molecules undergo hydrolysis, it forms either monosaccharides, disaccharides or trisaccharides. The end products depends on the strength of enzymes used and the common enzymes are, α-Amylase, which produces the disaccharide maltose and the trisaccharide maltotriose.What is starch hydrolysis?
Starch hydrolysis test. This test is used to identify bacteria that can hydrolyze starch (amylose and amylopectin) using the enzymes a-amylase and oligo-1,6-glucosidase. These enzymes break the starch molecules into smaller glucose subunits which can then enter directly into the glycolytic pathway.How do you test for sucrose in a solution?
The Benedict's test heats a mixture of Benedict's reagent (a deep-blue alkaline solution) and sugar. If a reducing sugar is present, the reagent changes color: from green to dark red or rusty-brown, depending on the quantity and type of sugar. If you add a non-reducing sugar, like sucrose, the reagent remains blue.Why is sucrose easily hydrolyzed in an acid solution?
Sucrose and fondant. Sucrose is very easily hydrolyzed, even by very weak acids, though the addition of water to the sugar molecule cannot be brought about by mixing sugar and water, but may be brought about by enzymes as well as acids. Hydrolysis takes place more rapidly if the solution is heated.Can sucrose be oxidized?
Because sucrose is not reducing sugar. In order for sugar to be oxidised by Benedict reagent it has to contain aldehyde group or has to isomerise in the solution to get one. This is possible for all monosaccharides. Lactose has one of its formerly hemiacetal oxygen free and therefore, it can form aldehyde.