What type of bonding do noble gases have?
.
Similarly one may ask, can noble gasses bond?
The Chemical Properties The full valence electron shells of these atoms make noble gases extremely stable and unlikely to form chemical bonds because they have little tendency to gain or lose electrons. Although noble gases do not normally react with other elements to form compounds, there are some exceptions.
Also Know, why do heavier noble gases form compounds? Chemists originally believed that the noble gases could not form compounds , because their full valence shell of electrons made them chemically stable and unreactive. But the heavier noble gases have more electron shells than the lighter ones. Since then, many other noble gas compounds have been synthesized.
Also asked, which type of bond is present in Xe molecule?
In xenon clathrates the xenon molecules occupy the gaps in the open hydrogen-bonded networks of the water molecules. There is no conventional chemical bonding involved in such clathrates, as in the case of halogen clathrates.
Why would covalent bonding be similar to a noble gas status?
1 Answer. Covalent bonding occurs when atoms share valence electrons to form a chemical bond. Atoms of nonmetals form covalent bonds by sharing electrons in order to achieve an octet of valence electrons, like the noble gases, except for helium, which only has two valence electrons.
Related Question AnswersDo noble gases have electronegativity?
Electronegativity is the ability of an atom to gain an electron. Since the noble gases already have eight electrons in their outer shells, they dont wan't to attract any more. Since electronegativity measures the amount of attraction between an atom and an electron, noble gases do not have electronegativity.Are noble gases flammable?
The main properties of the noble gases include: they have low densities. they are inert, so they are not flammable.Can noble gases be ionized?
The heavier noble gases have more electron shells than the lighter ones. Hence, the outermost electrons are subject to a shielding effect from the inner electrons that makes them more easily ionized, since they are less strongly attracted to the positively charged nucleus.Why halogens are so reactive?
Halogens are highly reactive, and they can be harmful or lethal to biological organisms in sufficient quantities. This reactivity is due to high electronegativity and high effective nuclear charge. Halogens can gain an electron by reacting with atoms of other elements. Fluorine is one of the most reactive elements.Why do noble gases not bond?
The Chemical Properties The full valence electron shells of these atoms make noble gases extremely stable and unlikely to form chemical bonds because they have little tendency to gain or lose electrons. Although noble gases do not normally react with other elements to form compounds, there are some exceptions.Can noble gases form ionic bonds?
This is called Ionic Chemical Bonding. But that's a different topic. So, for noble gases, they already have their outermost shells filled completely so they need not donate or accept any electrons. So, they don't need to form ions.Are noble gases dangerous?
Noble gases are very stable chemicals that do not react easily with other chemicals and are not flammable. These gases are not very toxic but they can be harmful in large amounts. Noble gases are asphyxiants. Asphyxiant gases replace oxygen in the air so there is less of it to breathe.What makes a noble gas noble?
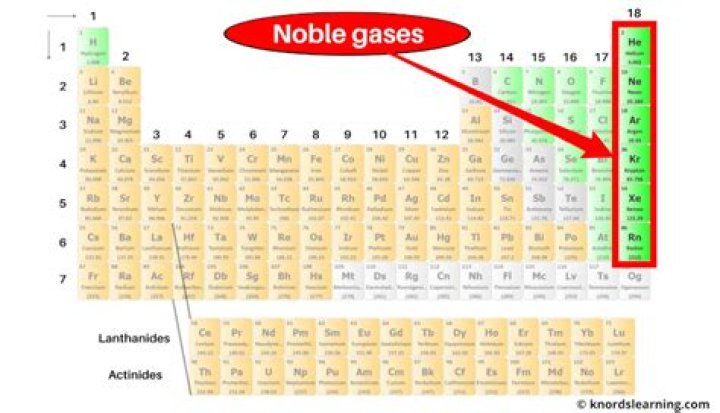
Noble gas. The noble gases are the chemical elements in group 18 of the periodic table. They are the most stable due to having the maximum number of valence electrons their outer shell can hold. Therefore, they rarely react with other elements since they are already stable.Are noble gases stable?
Noble gas. The noble gases are the chemical elements in group 18 of the periodic table. They are the most stable due to having the maximum number of valence electrons their outer shell can hold. Therefore, they rarely react with other elements since they are already stable.Are noble gases Monatomic?
Noble gases, most often found as monatomic gases, have completely filled outer electron shells, so have no inclination to react with other elements, thus very rarely forming compounds with other elements.Are there any compounds of helium?
Helium compounds. Helium is one of the most unreactive elements, so it was commonly believed that helium compounds do not exist at all. So at extremely low temperatures helium may form van der Waals molecules. Repulsive forces between helium and other atoms may be overcome by high pressures.Do noble gases react with anything?
Noble gas. The noble gases are the chemical elements in group 18 of the periodic table. They are the most stable due to having the maximum number of valence electrons their outer shell can hold. Therefore, they rarely react with other elements since they are already stable.Why are there no compounds of helium?
Helium is the second most abundant element in the universe . but there are no known compounds of helium because IT IS A NOBLE GAS. all the elements in the universe except noble gases take part in chemical reactions and form compounds to gain stable electronic configuration like noble gases.What is argon used for?
It is used to fill incandescent and fluorescent light bulbs to prevent oxygen from corroding the hot filament. Argon is also used to form inert atmospheres for arc welding, growing semiconductor crystals and processes that require shielding from other atmospheric gases.Why is helium not a noble gas?
At the top of the noble gases is little helium (He), with a shell that is full with only two electrons. The fact that their outer shells are full means they are quite happy and don't need to react with other elements. In fact, they rarely combine with other elements. That non-reactivity is why they are called inert.Why is Xenon the most reactive noble gas?
Why? Noble gases are generally unreactive, but XENON compounds are reactive. It is so because the electrons in Xe are much less strongly bound to the atom , owning to it's large size.What makes a gas inert?
An inert gas is a gas that has extremely low reactivity with other substances. The noble gases—helium, argon, neon, xenon, krypton, radon, and element 118 (Uuo)—exist in their elemental form and are found in Group 18 of the periodic table. Inert gases have many practical uses because of their low reactivity.What are 3 types of covalent bonds?
The three types as mentioned in the other answers are polar covalent, nonpolar covalent, and coordinate covalent. The first, polar covalent, is formed between two nonmetals that have a difference in electronegativity. They share their electron density unevenly.What are 5 examples of covalent bonds?
Examples of Covalent Bond:- Water. An example is water. Water consists of a covalent bond containing hydrogen and oxygen bonding together to make H2O.
- Diamonds. A diamond is an example of Giant Covalent bond of carbon. A diamond has a giant molecular structure.
- Vulcanized rubber. Another example is vulcanized rubber.