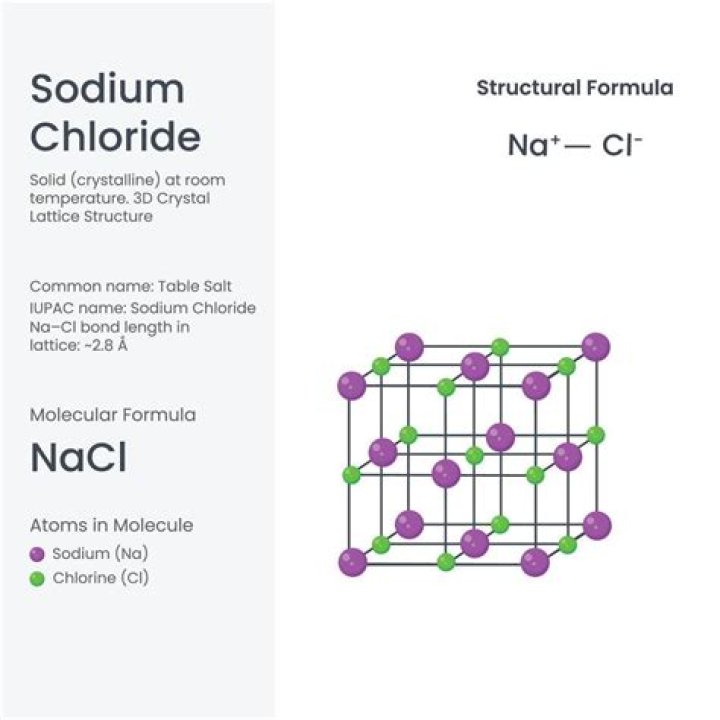
A combination reaction is one in which two or more substances (the reactants) are combined directly to form a single product (the product). An example is the reaction in which sodium (Na) combines with chlorine (Cl 2 ) to form sodium chloride, or table salt (NaCl)..
Just so, what is the formation of sodium chloride?
Sodium chloride is formed when sodium atoms interact with chlorine atoms. When this occurs, sodium will donate an electron (which is a negatively-charged particle) to chlorine. This makes sodium slightly positive and chlorine slightly negative. So then, sodium ions will attract chloride ions and form an ionic bond.
Likewise, what kind of chemical reaction does the chemical equation sodium chlorine → sodium chloride represent? synthesis reaction
One may also ask, is the formation of NaCl spontaneous?
Salt dissolves in water until it reaches its solubility limit. At the limit, the ions are concentrated enough to reverse the process which creates a dynamic equilibrium between the solid salt and the ions in solution. So the spontaneous solution process occurs only as long as the entropy change favors dissociation.
What are the uses of sodium chloride?
Sodium chloride is the chemical name for salt. Sodium is an electrolyte that regulates the amount of water in your body. Sodium also plays a part in nerve impulses and muscle contractions. Sodium chloride is used to treat or prevent sodium loss caused by dehydration, excessive sweating, or other causes.
Related Question Answers
What is the chemical name of sodium chloride?
Sodium chloride is one of the most well-known and widely used chemicals, also known as table salt. Formula and structure: The chemical formula of sodium chloride is NaCl and its molar mass is 58.44 g/mol. It is an ionic compound consisting of a sodium cation (Na+) and a chloride anion (Cl-).What is the purpose of sodium chloride IV?
Sodium Chloride IV Infusion is used for treatment of fluid loss and to restore sodium chloride balance. It is used in the treatment of patients who are unable to take fluids and nutrients by mouth. It is also used for dilution of other medicines before injection into the body.Is NaCl an acid or base?
NaCl is formed by the reaction of HCl and NaOH. Both are strong acids and bases. When a strong acid and a strong base react together the resultant is salt and water. Therefore NaCl is a salt.Why is the formula for sodium chloride NaCl?
So then, sodium ions will attract chloride ions and form an ionic bond. By the way, chloride is the term used to designate the anion form of chlorine. The chemical formula for sodium chloride is NaCl, which means that for every sodium atom present, there is exactly one chloride atom.What does sodium chloride do for the eyes?
Hypertonic sodium chloride eye ointment and eye drops are used to draw water out of a swollen cornea. The extra water in the cornea can cause your vision to be poor. You may also see halos or rings around point light sources. The cornea is the clear cover over the colored part of your eye.What is the product of NaCl h2o?
Nacl + H2O →product? Sodium chloride is react with water molecule to form the products are called sodium hydroxide and Hydrochloric acid.Why is NaCl not NaCl2?
The difference between NaCl and NaCl2 is that NaCl is the chemical formula for sodium chloride while NaCl2 does not exist. This is because, sodium is univalent and chlorine is also univalent, so during chemical combination between the two elements, there'll be interchanging of the valency.What is Delta H?
In chemistry, the letter "H" represents the enthalpy of a system. Enthalpy refers to the sum of the internal energy of a system plus the product of the system's pressure and volume. The delta symbol is used to represent change. Therefore, delta H represents the change in enthalpy of a system in a reaction.Why does the temperature decrease when NaCl is dissolved in water?
The process of dissolving is endothermic when less energy is released when water molecules “bond” to the solute than is used to pull the solute apart. Because less energy is released than is used, the molecules of the solution move more slowly, making the temperature decrease.Why is the formation of NaCl exothermic?
NaCl exists as a crystal lattice of sodium and chlorine ions, where the extra electron from a sodium atom fills in the "hole" needed to complete a chlorine atom's outer electron shell. While the gain of an electron by a nonmetal is usually exothermic (energy is released when the nonmetal gains a full octet).Why is dissolving NaCl endothermic?
This means just slightly more energy must be put into the solution than is released back into the solution; therefore dissolving table salt in water is endothermic. More energy is released into the solution than is required to pull apart the ions; therefore dissolving sodium hydroxide in water is exothermic.Is NaCl a solid?
The isolated Na+ and Cl- ions in the gas phase then come together to form solid NaCl.Is dissolving NaCl endothermic or exothermic?
Answer and Explanation: Dissolving salt in water is endothermic. This means that when salt is dissolved in water the temperature of the solution is often a little lower thanWhat happens when you mix salt and chlorine?
Sodium + Chlorine: When a sodium atom transfers an electron to a chlorine atom, forming a sodium cation (Na+) and a chloride anion (Cl-), both ions have complete valence shells, and are energetically more stable. The reaction is extremely exothermic, producing a bright yellow light and a great deal of heat energy.What does spontaneous mean in chemistry?
A spontaneous reaction is a reaction that occurs in a given set of conditions without intervention. Spontaneous reactions are accompanied by an increase in overall entropy, or disorder. If the Gibbs Free Energy is negative, then the reaction is spontaneous, and if it is positive, then it is nonspontaneous.What type of reaction is Na cl2 NaCl?
is an example of a synthesis reaction. In your case, sodium metal, Na , reacts with chlorine gas, Cl2 , to form sodium chloride, NaCl .What type of reaction is Na h2o?
displacement reaction
What type of reaction takes place between a chloride and water?
Reactions with water As an approximation, the simple ionic chlorides (sodium and magnesium chloride) just dissolve in water. The other chlorides all react with water in a variety of ways described below for each individual chloride. The reaction with water is known as hydrolysis.How do you make sodium chloride formula?
NaCl