What is the structure of aluminium chloride?
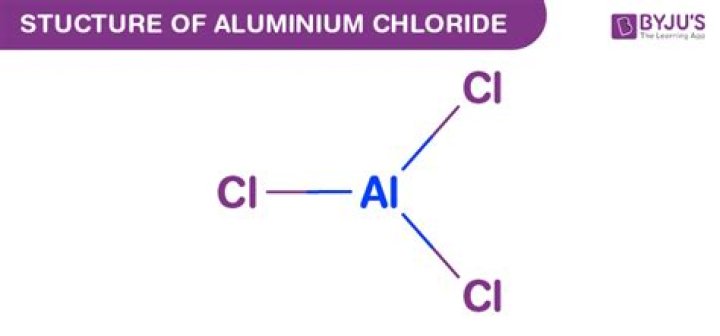
What is the structure of aluminium chloride?
AlCl₃
Aluminium chloride/Formula
How many electrons does AlCl3 have?
In AlCl3 Al have 3 valence electron and Cl have 7 valence electron, total electrons => 3 + 7*3 => 3 + 21 => 22.
How many electron orbital shells does aluminum have?
List of elements with electrons per shell
| Z | Element | No. of electrons/shell |
|---|---|---|
| 10 | Neon | 2, 8 |
| 11 | Sodium | 2, 8, 1 |
| 12 | Magnesium | 2, 8, 2 |
| 13 | Aluminium | 2, 8, 3 |
What is the charge an aluminum ion?
The charge of an aluminum ion is typically 3+. This is because the element’s atomic number is 13, reflecting the fact that it has 13 electrons and 13 protons. The valence shell of aluminum has three electrons, and per the octet rule, these three electrons are lost resulting in just 10 electrons and 13 protons.
Is the atomic number for aluminum?
13
Aluminium/Atomic number
What is dimeric structure of AlCl3?
This change in structure is related to the lower density of the liquid phase (1.78 g/cm3) vs solidaluminium trichloride (2.48 g/cm3). Al2Cl6 dimersare also found in the vapour phase. At higher temperatures, the Al2Cl6 dimers dissociate into trigonal planar AlCl3, which is structurally analogous to BF3.
Is Aluminium chloride a precipitate?
Aqueous solutions behave similarly to other aluminium salts containing hydrated Al3+ ions, giving a gelatinous precipitate of aluminium hydroxide upon reaction with dilute sodium hydroxide: AlCl3 + 3 NaOH → [Al(OH)3] + 3 NaCl.
Is aluminum chloride a precipitate?
Aqueous solutions of aluminum chloride and sodium hydroxide are mixed, forming the precipitate aluminum hydroxide.
How many electrons does aluminum have?
thirteen electrons
Electron Configuration of Aluminum To find the electron configuration of an atom, you first need to know the number of electrons that it has. Since aluminum’s atomic number is thirteen, it has thirteen electrons.



