Platinum is a solid at room temperature..
Consequently, what is the state of matter of platinum?
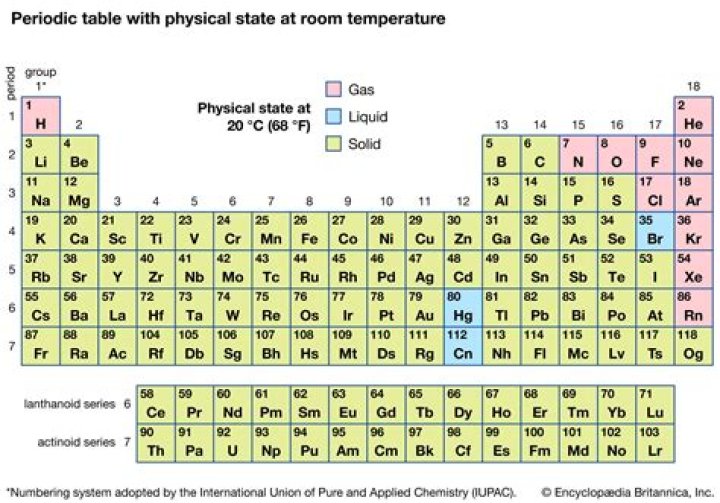
Elements can be classified based on their physical states (States of Matter) e.g. gas, solid or liquid. This element is a solid. Platinum is classified as a "Transition Metal" which are located in Groups 3 - 12 of the Periodic Table.
Subsequently, question is, what elements are solid at room temperature? Hydrogen, nitrogen, oxygen, fluorine, and chlorine are gases at STP.
Likewise, how is platinum formed in nature?
Platinum in the Earth's crust originates from ultra-mafic igneous rocks. It can therefore be associated with rocks like chromite and olivine. In nature, pure platinum is unknown and well formed crystals are very rare. Platinum is typically found as nuggets and grains.
Can Platinum be a liquid?
when liquid (at m.p. ) 2800 m/s (at r.t. ) Platinum is a chemical element with the symbol Pt and atomic number 78. It is a dense, malleable, ductile, highly unreactive, precious, silverish-white transition metal.
Related Question Answers
What color is pure platinum?
white
How can you tell if its real platinum?
Platinum is a very rare and expensive metal. Therefore, all platinum jewelry will be marked to show authenticity. Look for the words “Platinum," “PLAT," or “PT” followed or preceded by the numbers “950” or “999." These numbers refer to the purity of the platinum, with “999” as the most pure.Is Platinum toxic to humans?
Health effects of platinum Platinum is a noble metal. The concentrations of platinum in the soil, water and air are very minimal. Finally, a danger of platinum is that it can cause potentiation of the toxicity of other dangerous chemicals in the human body, such as selenium.Where is platinum formed?
Platinum is found in its pure native form or in platiniridium, a natural alloy of platinum and iridium. Platinum is present in thin sulfide layers in certain mafic igneous bodies and is mined in Canada, Russia, South Africa, the USA, Zimbabwe and Australia.What is platinum used for?
Platinum is widely used as a catalyst for chemical reactions. The most important use of platinum is in vehicles, as a catalytic converter, facilitating the complete combustion of unburned hydrocarbon passing through the exhaust. Platinum is used in jewelry, decoration and dental work.What state of matter is iron?
The Atomic Number of this element is 26 and the Element Symbol is Fe. Elements can be classified based on their physical states (States of Matter) e.g. gas, solid or liquid. This element is a solid. Iron is classified as a "Transition Metal" which are located in Groups 3 - 12 of the Periodic Table.Is platinum a good conductor?
It is one of the densest metal elements—almost twice as dense as lead—and very stable, giving the metal excellent corrosion resistant properties. A good conductor of electricity, platinum is also malleable (able to be formed without breaking) and ductile (able to be deformed without losing strength) .How much platinum is there?
According to the U.S. Geological Survey, the world mined about 192 tons of platinum in 2011, so at today's rate, it would take about 100 years to get it out of the ground. But as we said, if the president decided he wanted Treasury to produce a $1 trillion platinum coin, he just has to say so.What rock is platinum found in?
igneous rocks
How strong is Platinum?
Platinum is Stronger and More Durable Though both gold and platinum are strong and durable precious metals, platinum is the stronger and more durable of the two. For example, the prongs holding the center stone of a platinum engagement ring are less likely to break then those of a gold engagement ring.Is Platinum better than gold?
The biggest advantage of white gold over platinum is certainly the cost. Platinum is more expensive because it is rarer and mined much less than gold. Also, platinum is more dense than gold, so the same ring will weigh significantly more in platinum than in gold (and precious metals are priced by weight).What products contain platinum?
The versatile platinum group metals (PGMs)—platinum, palladium, rhodium, iridium, ruthenium and osmium—can be found in products we use every day, such as catalytic converters, drugs and medical devices, and many, many electronic devices.How do you know if an element is solid at room temperature?
Physical States — Melting Points, Boiling Points, and Densities. In the periodic table above, black squares indicate elements which are solids at room temperature (about 22ºC)*, those in blue squares are liquids at room temperature, and those in red squares are gases at room temperature.What do halogens look like?
The halogens become darker as you go down the group. Fluorine is very pale yellow, chlorine is yellow-green, and bromine is red-brown. Iodine crystals are shiny purple - but easily turn into a dark purple vapour when they are warmed up.Why are Group 17 called halogens?
Group 17 elements are called halogens because halogen is a Greek word which means 'salt producing'. Halogens include fluorine, chlorine, bromine, iodine and astatine. They all are non-metals. They react with metals to form compounds called salts.What state is Group 1 at room temperature?
The alkali metals are solids at room temperature (except for hydrogen), but have fairly low melting points: lithium melts at 181ºC, sodium at 98ºC, potassium at 63ºC, rubidium at 39ºC, and cesium at 28ºC.How many halogens are there?
Types of Halogens Depending on who you ask, there are either 5 or 6 halogens. Fluorine, chlorine, bromine, iodine, and astatine definitely are halogens.Why are halogens so reactive?
Halogens are highly reactive, and they can be harmful or lethal to biological organisms in sufficient quantities. This reactivity is due to high electronegativity and high effective nuclear charge. Halogens can gain an electron by reacting with atoms of other elements. Fluorine is one of the most reactive elements.