What is the pKa of nh3?
What is the pKa of nh3?
38
The pKa of ammonia itself is 38, which measures the equilibrium constant for dissociation of NH3 to give its conjugate base, NH2(-) and H+.
What is the pKa of H2Se?
Hydrogen selenide
| Names | |
|---|---|
| Acidity (pKa) | 3.89 |
| Conjugate acid | Selenonium |
| Conjugate base | Selenide |
| Structure |
Is H2Se a strong acid or base?
In binary acids such as H2S and H2Se, the H–Se bonds is longer than the H–S bonds as Se is larger than S. The H–Se bond is therefore weaker than the H–S bond and H2Se is thus a stronger acid than H2S.
Is HBr or H2Se a stronger acid?
Yes, hydrogen bromide (HBr) is stronger than hydrogen selenide (H2Se).
What is structure of NH3?
The ammonia molecule has a trigonal pyramidal shape with the three hydrogen atoms and an unshared pair of electrons attached to the nitrogen atom. It is a polar molecule and is highly associated because of strong intermolecular hydrogen bonding.
What does a pKa of 8 mean?
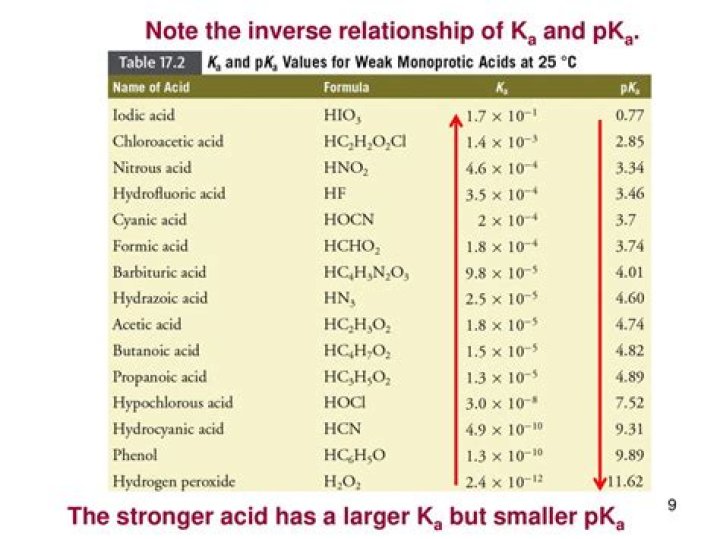
Remember that a pKa table ranks molecules in order of their acidity, from strongly acidic (e.g. HCl with pKa of –8) to weakly acidic (e.g. methane, pKa of ~50). We apply the following principle to acid-base reactions: A stronger acid will tend to react with a stronger base to produce a weaker acid and a weaker base.
Is AsH3 stronger than H2Se?
AsH3 is a stronger acid than HBr because As is larger than Br. H2SO4 is a stronger acid than H3PO4 because S is more electronegative than P. H2S is a stronger acid than H2Se because S is more electronegative than Se.
What is the pKa of H20?
Using the latter standard state, the equilibrium constant for the water/methoxide reaction ranges from 4 to 70, showing water to be a stronger acid than methanol, and justifying the use of the thermodynamically correct value, pKa(H2O) = 14.00.
Why h2o is less acidic than H2Se?
H2S is less acidic than H2Te because on moving down the group the size of the atom increases and ionization enthalpy decreases. This helps to dissociate the bond easily. But in case of H2Sthe bond dissociation is less and hence acidity is lower.
Is Bro or BrO2 a strong base?
The answer is A : BrO2− . The strength of the conjugate base can be predicted from the strength of the acid.
What is the 3d structure of NH3?
NH3 Ammonia Ammonia has 4 regions of electron density around the central nitrogen atom (3 bonds and one lone pair). These are arranged in a tetrahedral shape. The resulting molecular shape is trigonal pyramidal with H-N-H angles of 106.7°.
What is NH3 geometry?
ammonia. The ammonia molecule has a trigonal pyramidal shape with the three hydrogen atoms and an unshared pair of electrons attached to the nitrogen atom. It is a polar molecule and is highly associated because of strong intermolecular hydrogen bonding.