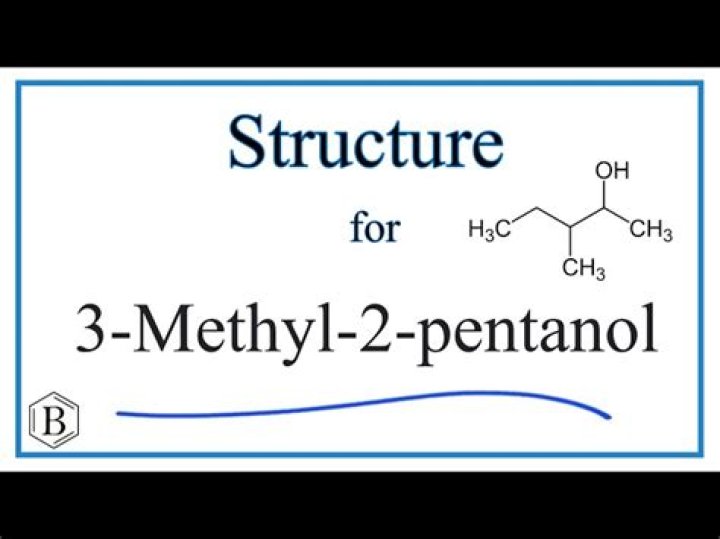
What is the organic product formed from the dehydration of 3 Methyl 2 Pentanol?
.
Correspondingly, what is the product of dehydration of 3 Methyl 2 Pentanol?
Introduction The acid-catalyzed dehydration of 3-methyl-3-pentanol gives a product mixture that contains the isomeric alkenes 3-methyl-2-pentene and 2-ethyl-1-butene.
Secondly, what is the major product formed in the acid catalyzed reaction of 2 methyl 2 pentene with water? Answer and Explanation: When 2-methyl-2-pentene reacts with sulfuric acid in presence of water, hydration occurs to form 2-methylpentan-2-ol as the major product.
Subsequently, one may also ask, what is the major product that results from the dehydration of 2 Methyl 2 Pentanol?
Answer and Explanation: The dehydration of alcohol will result in the formation of an olefin(alkene). The dehydration of 2-methyl-2-pentanol forms one major and one minor The dehydration of alcohol will result in the formation of an olefin(alkene).
What compound is produced when 3 Pentanol is subjected to controlled oxidation?
iii) gave cyclohexene oxide.
Related Question Answers