What is the correct order of stability of carbocations?
What is the correct order of stability of carbocations?
The correct order for the carbocation stability is: III > I > II.
What is the stability order for a carbocation from most stable to least?
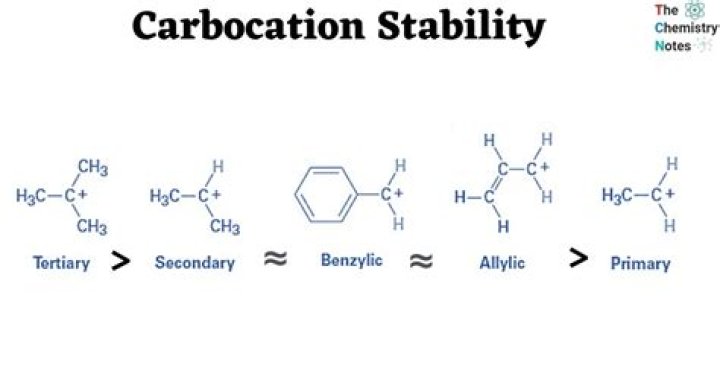
Increasing the number of adjacent carbon atoms: methyl (least stable carbocation) < primary < secondary < tertiary (most stable carbocation)
Which is the most stable carbocation formula?
The most stable carbocation is t-alkyl carbocation because the order of stability of alkyl carbocation is t-alkyl >s-alkyl> p-alkyl *> CH3 carbocation.
What is the order of stability of Carbanion?
The stability order of carbanion decreases, while moving from primary to the tertiary anion, due to increase intensity of negative charge on central carbon of tertiary anion.
Which is the correct order of stability?
I > III > IV > II.
Which is correct order of stability of free radical?
Stability Of Free Radicals Increases In The Order Methyl < Primary < Secondary < Tertiary.
Why are carbocations unstable?
Explanation: A carbocation is an organic molecule, an intermediate, that has a carbon atom bearing a positive charge and three bonds instead of four. Since the charged carbon atom does not satisfy the octet rule, it is unstable and therefore highly reactive.
How do carbocations rearrange?
There are two types of carbocation rearrangements: a hydride shift and an alkyl shift. Once rearranged, the resultant carbocation will react further to form a final product which has a different alkyl skeleton than the starting material.
What is carbocation stability?
Alkyl groups – methyl, ethyl, and the like – are weak electron donating groups, and thus stabilize nearby carbocations. What this means is that, in general, more substituted carbocations are more stable: a tert-butyl carbocation, for example, is more stable than an isopropyl carbocation.
What makes a stable carbocation?
The three factors that determine carbocation stability are adjacent (1) multiple bonds; (2) lone pairs; and (3) carbon atoms. An adjacent π bond allows the positive charge to be delocalized by resonance. Resonance delocalization of the charge through a larger π cloud makes the cation more stable.
What makes Carbocations more stable?
What is the order of stability of free radicals?



