What is the conjugate acid for NH3?
What is the conjugate acid for NH3?
NH4+
NH4+ is the conjugate acid of NH3.
What is the conjugate acid and base for NH3 respectively?
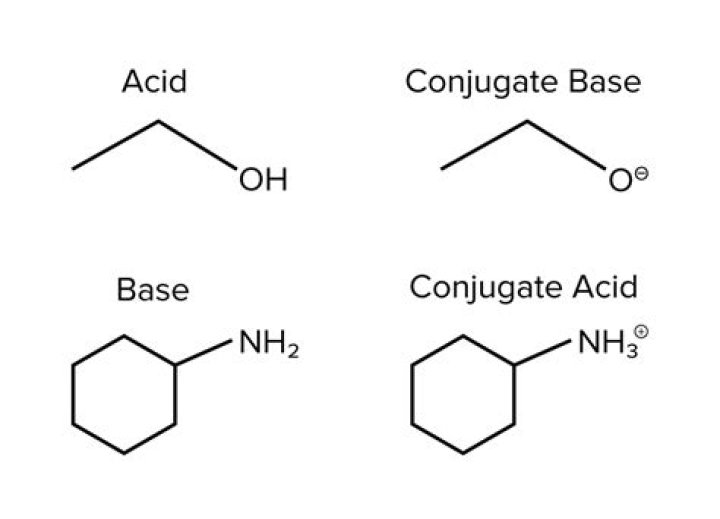
Ammonia or NH3 is a base. It accepts a proton to give its conjugate and NH4+. Similarly, NH4+ loses a proton to give conjugate base NH3. Therefore, NH4+ is the conjugate acid of ammonia.
What is NH2 conjugate acid?
The conjugate acid of amide ion (NH2−) is ammonia, NH3. When a proton is added to a base, we get conjugate acid.
Is ammonia a conjugate acid or base?
In this reaction, ammonia is the conjugate base and accepts a proton from water, where it becomes the conjugate acid ammonium. You can show that the lone pair of electrons on the nitrogen atom reacts with the hydrogen on water molecules, giving an ammonium ion and a hydroxide ion.
Which species are the conjugate acid and conjugate base of NH3?
The conjugate acid of NH3 is NH+4 , ammonium ion; the conjugate base is NH−2 , amide ion.
Why is NH3 a Bronsted base?
Explanation: A Brønsted-Lowry base is a substance that behaves as a proton acceptor. The NH3 is accepting a proton from the HCl , so it is behaving as a Brønsted-Lowry base.
Are NH3 and NH2 conjugate pairs?
In this case, NH3 is considered the acid since it donates a proton, so the conjugate base would be NH2-.
What is NH2 and NH3 conjugate acid?
Write the conjugate acids for the following Brönsted bases: NH2–, NH3 and HCOO–….Solution.
| Bronsted base | Conjugate acid |
|---|---|
| NH | NH |
| NH | NH |
| HCOO | HCOOH |
Is NH3 acid or base?
Ammonia, NH3, is a Lewis base and has a lone pair. It will donate electrons to compounds that will accept them. Donation of ammonia to an electron acceptor, or Lewis acid. There may be anionic or neutral Lewis bases.
What is conjugate acid base pair name the conjugate base of ammonia with equation?
Acid + Base ⇌ Conjugate Base + Conjugate Acid….Table of acids and their conjugate bases.
| Acid | Conjugate base |
|---|---|
| NH + 4 Ammonium ion | NH3 Ammonia |
| H2O Water (pH=7) | OH− Hydroxide ion |
What is the conjugate acid of hpo42 −?
When we add a proton, we must conserve both mass and charge, we add H+ to HPO2−4 so the conjugate acid is simply H2PO−4 .
Why is NH3 a Bronsted Lowry acid?
Because the water molecule donates a hydrogen ion to the ammonia, it is the Brønsted-Lowry acid, while the ammonia molecule—which accepts the hydrogen ion—is the Brønsted-Lowry base. Thus, ammonia acts as a base in both the Arrhenius sense and the Brønsted-Lowry sense.