What is the charge of the cation in MnO2?
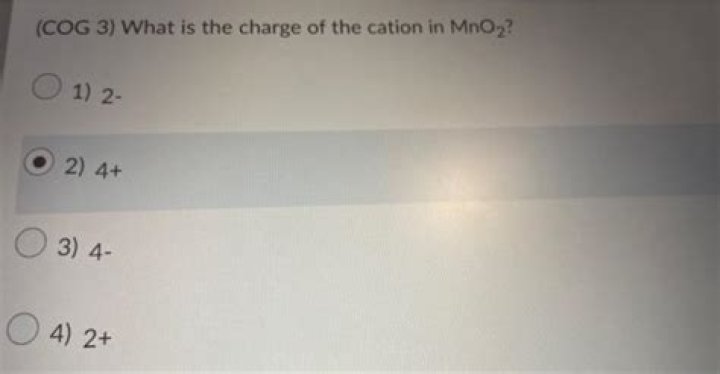
What is the charge of the cation in MnO2?
-2
Let x be the oxidation number of Mn in MnO2. Since the overall charge on the complex is 0, the sum of oxidation states of all elements in it should be equal to 0. This compound, called manganese dioxide, also contains two oxygen ions, each with an oxidation number of -2.
Is MnO2 ionic?
The name of MnO2 M n O 2 is manganese(IV) oxide and it is an ionic compound. Name of the nonmetal with its ending changed to “ide”.
What is the oxidation state for Mn in MnO2?
We’re being asked to determine the oxidation state of manganese in MnO2 (manganese (IV) oxide). Since Mn2O3 is neither a peroxide or superoxide, the oxidation state of each oxygen atom is –2. The oxidation state of Mn in MnO2 is +4.
How is MnO2 formed?
How is manganese dioxide formed? By oxidation of the elemental manganese: elemental manganese reacts with oxygen in the environment to form MnO2. Because of this reaction elemental manganese does not exist in nature – it is usually found as manganese dioxide in nature.
What is the oxidation state of each V in V2O5?
Vanadium, on the other hand, at its +5 oxidation state (V2O5), acts as a potent neurotoxic agent.
What compound is MnO2?
Manganese oxide
Manganese(IV) oxide
Manganese(IV) oxide/IUPAC ID
What type of oxide is MnO2?
Manganese dioxide is a manganese molecular entity with formula MnO2. It is a manganese molecular entity and a metal oxide.
Which of the following anions is polyatomic?
| Symbols and Names of Some Common Polyatomic Ions and One Molecule | ||
|---|---|---|
| NH4+ | ammonium ion | cyanide ion |
| HSO4- | hydrogen sulfate ion | thiocyanate ion |
| SO32- | sulfite ion | carbonate ion |
| NO3- | nitrate ion | hydrogen carbonate |
What is the oxidation number CR in cr2o?
+ 6
The sum of the oxidation numbers in Cr2O72-, a polyatomic ion, is -2, the charge of the ion. There are seven oxygen atoms, so the total charge is − 2⋅7 = − 14. So, a chromium atom here has an oxidation number of + 6.
Is MnO2 a neutral oxide?
MnO has the lowest oxidation state, so it is the most basic and finally Mn2O7 (which is actually acidic). So, basically MnO2 is weakly acidic and mildly basic and can somewhat be classified as amphoteric.
What Colour is MnO2?
Manganese dioxide (MnO2) is an inorganic compound. It is a black to brown-colored material that occurs naturally as the mineral pyrolusite (see Figure 1).