What is the angle of a bent molecule?
What is the angle of a bent molecule?
In bent molecules, the bond angle is slightly less than 120∘ . This is because lone pairs take up more room than single bonds do. Therefore, the lone pair in a bent molecule takes up more room than the 3rd bond in a trigonal planar molecule does, thereby reducing the angle to slightly less than 120∘ .
What is a bent shape in chemistry?
The shape of the orbitals is tetrahedral. Two of the orbitals contain lone pairs of electrons. The two atoms connected to the central atom form a molecule with a bent shape.
Is bent sp2?
For sp2 hybridized central atoms the only possible molecular geometry is trigonal planar. If there are only three bonds and one lone pair of electrons holding the place where a bond would be then the shape becomes trigonal pyramidal, 2 bonds and 2 lone pairs the shape is bent.
What determines bent or linear?
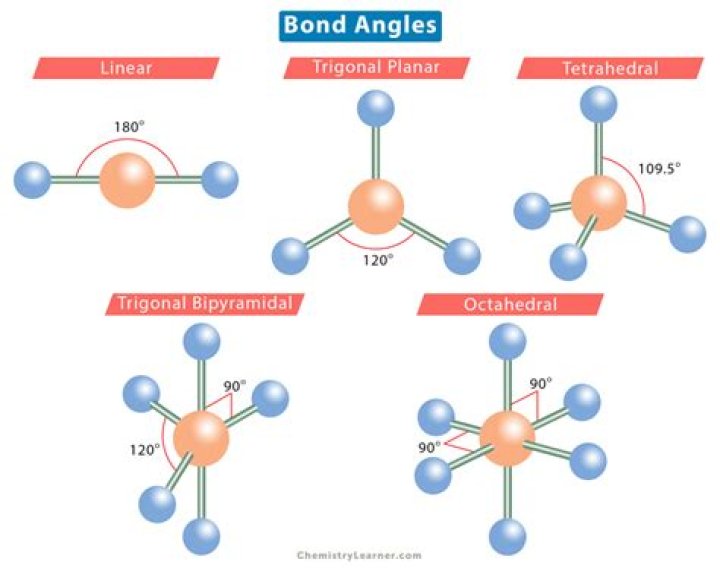
1st remember the names: The names can be determined by the shape and angle of the molecule. Linear = is just a line of atoms with a 180° angle. Notice that it’s 2 or 3 atoms total. Bent = Linear but bent due to the Lone Pairs that it contains, the more Lone Pairs the greater the bent and the smaller the degree.
Why is a molecule bent?
The reason water has a bent shape is that the two lone pair of electrons are on the same side of the molecule. This repulsion of the lone pairs of electrons on the oxygen atom causes the bond of the hydrogen to the oxygen to be pushed downward (or upward, depending on your point of view).
Is H2S bent?
H2S molecular geometry is bent. H2S electron geometry is tetrahedral. The total valence electron available for drawing the lewis structure of H2S is 8. The lone pair in on the central atom is 2 according to the lewis structure of SH2/H2S.
What has bent shape?
Water (H2O) is an example of a bent molecule, as well as its analogues. The bond angle between the two hydrogen atoms is approximately 104.45°….
| Bent molecular geometry | |
|---|---|
| Coordination number | 2 |
| Bond angle(s) | 90°<θ<120° |
| μ (Polarity) | >0 |
What causes a bent molecule?
BENT (ANGULAR or V-SHAPED) It can be imagined that a linear molecule with two atoms attached to a central atom is altered when electrons are added to the top of the central atom. The repulsion caused by the addition of these extra electrons causes the molecule to become bent.
Is o3 molecule bent?
Ozone has three electron groups around the central oxygen, so has the trigonal planar electron geometry. The molecular geometry is bent: The Effect of Lone Pairs (contd.) With four electron groups (tetrahedral electron geometry), there can be one or two lone pairs around the central atom.
Is so2 molecule bent?
Carbon dioxide is linear, while sulphur dioxide is bent (V-shaped). In sulphur dioxide, as well as the two double bonds, there is also a lone pair on the sulphur. To minimise repulsions, the double bonds and the lone pair get as far apart as possible, and so the molecule is bent.
What is the difference between linear and bent?
The key difference between linear and bent molecules is that linear molecules have atoms bonded to each other, forming a straight molecule, whereas bent molecules have the atoms arranged in a bend-shape with an angle.



