What is the 1st and 2nd ionisation energy?
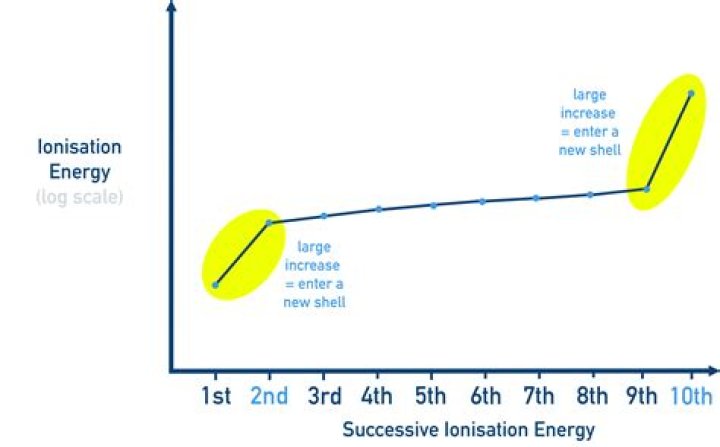
What is the 1st and 2nd ionisation energy?
First ionization energy: The energy needed to remove the outermost, or highest energy, electron from a neutral atom in the gas phase. Second ionization energy: The energy it takes to remove an electron from a 1+ ion (meaning the atom has already lost one electron and now removing the second).
What is 2nd ionisation potential?
Second ionization energy can be defined as the removal of one mole of electron from the one mole of monovalent gaseous cation of the element as after first ionization process, the metal forms an isolated monovalent gaseous cation due to loss of one electron from the atom.
What is ionization energy easy definition?
17 hours ago
ionization energy, also called ionization potential, in chemistry and physics, the amount of energy required to remove an electron from an isolated atom or molecule. Among the chemical elements of any period, removal of an electron is hardest for the noble gases and easiest for the alkali metals.
What is the difference between the 1st ionization energy the 2nd ionization energy?
The first ionization energy is the energy it takes to remove an electron from a neutral atom. The second ionization energy is the energy it takes to remove an electron from a 1+ ion. (That means that the atom has already lost one electron, you are now removing the second.)
What is the second ionization energy of sodium?
This is the energy per mole necessary to remove electrons from gaseous atoms or atomic ions. The first molar ionization energy applies to the neutral atoms….1st–10th ionisation energies.
| Symbol | Na |
|---|---|
| Name | sodium |
| 2nd | 4562 |
| 3rd | 6910.3 |
| 4th | 9543 |
What is the second ionization energy of MG?
This is the energy per mole necessary to remove electrons from gaseous atoms or atomic ions. The first molar ionization energy applies to the neutral atoms….1st–10th ionisation energies.
| Number | 12 |
|---|---|
| Symbol | Mg |
| Name | magnesium |
| 1st | 737.7 |
| 2nd | 1450.7 |
How do you calculate second ionization energy?
The equation En=-Z2RH/n2 gives the energy (according to the Bohr model) of the energy levels available to an electron in any hydrogen-like atom. Calculate the second ionization energy of He(in J). The second ionization energy of He is the ionization energy of He+.
What is the second ionization energy of He+?
In helium’s case, Bohr’s equation predicts the same value for both the first, and the second ionization energies: 5276 kJ/mol, with only the second ionization energy being correct. For multi-electron atoms, you must replace Z with Zeff , the effective nuclear charge.
What is the second ionization energy of aluminum?
These tables list values of molar ionization energies, measured in kJ⋅mol−1….1st–10th ionisation energies.
| Number | 13 |
|---|---|
| Symbol | Al |
| Name | aluminium |
| 1st | 577.5 |
| 2nd | 1816.7 |
What is the second ionization energy of oxygen?
Oxygen atoms have 8 electrons and the shell structure is 2.6. The ground state electron configuration of ground state gaseous neutral oxygen is [He]….Ionisation Energies and electron affinity.
| Ionisation energy number | Enthalpy / kJ mol‑1 |
|---|---|
| 1st | 1313.94 |
| 2nd | 3388.67 |
| 3rd | 5300.47 |
| 4th | 7469.27 |