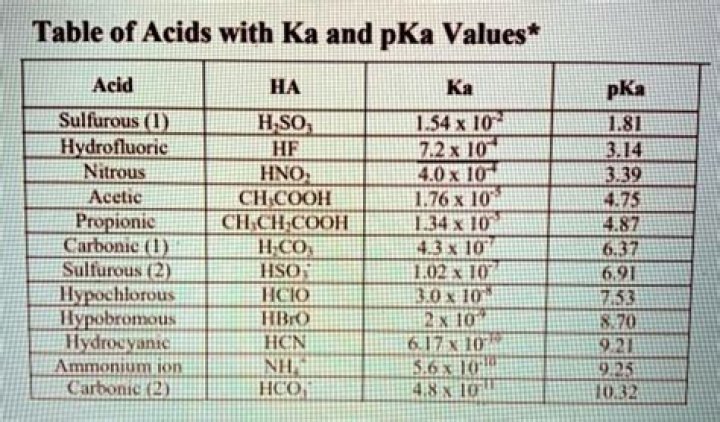
For example, the pKa of acetic acid is 4.8, whilethe pKa of lactic acid is 3.8..
People also ask, what are the Ka and pKa of acetic acid?
Acetic Acid Ka = 1.7×10^-5; – logKa = -4.76, therefore pKa = 4.76. So strongeracids have lower pKa's (or have higherKa's).
Also Know, what does a high pKa mean? The lower the pKa of a Bronsted acid, the moreeasily it gives up its proton. The higher the pKa ofa Bronsted acid, the more tightly the proton is held, and the lesseasily the proton is given up. Figure AB9.2. pKa cansometimes be so low that it is a negative number! High pKameans a proton is held tightly.
Likewise, people ask, what does the pKa tell you?
pKa is a property of a compound that tellsus how acidic it is. The lower the pKa, the stronger theacid. pH is a property of a particular solution that depends on theconcentrations and identities of the components. If the pH is lowerthan the pKa, then the compound will beprotonated.
What is acetic acid used for?
Acetic acid is an antibiotic that treatsinfections caused by bacteria or fungus. Acetic acid otic(for the ear) is used to treat infections in the earcanal.
Related Question Answers
Is HCl a strong acid?
A strong acid is an acid which iscompletely ionized in an aqueous solution. Hydrogen chloride(HCl) ionizes completely into hydrogen ions and chlorideions in water. Weak acids, like strong acids, ionizeto yield the H+ ion and a conjugate base. BecauseHCl is a strong acid, its conjugate base(Cl−) is extremely weak.What is the pKa of HCl?
More precisely, the acid must be stronger in aqueoussolution than a hydronium ion (H+), so strong acids havea pKa < -1.74. An example is hydrochloric acid(HCl), whose pKa is -6.3.What is the world's strongest acid?
The carborane superacids may be considered theworld's strongest solo acid, as fluoroantimonicacid is actually a mixture of hydrofluoric acid andantimony pentafluoride. Carborane has a pH value of-18.What is the Ka value for acetic acid?
The Ka value for acetic acid, CH3COOH(aq), is1.8x10^-5. Calculate the ph of a 2.80 M acetic acidsolution.What is pH and pKa?
pH and pKa The lower the pH, the higher the concentrationof hydrogen ions, [H+]. The lower the pKa, thestronger the acid and the greater its ability to donate protons.pH depends on the concentration of the solution. On theother hand, the pKa value is a constant for each type ofmolecule.What is pKa vs ka?
Ka is the acid dissociation constant. pKais simply the -log of this constant. Similarly, Kb is the basedissociation constant, while pKb is the -log of the constant. Theacid and base dissociation constants are usually expressedin terms of mole per liter (mol/L).What is the Ka of HCl?
| Ka | Acid |
| Large | Perchloric acid | HClO4 |
| 3.2 * 109 | Hydroiodic acid | HI |
| 1.0 * 109 | Hydrobromic acid | HBr |
| 1.3 * 106 | Hydrochloric acid | HCl |
Is hco3 an acid or base?
acid, H2CO3, and the weak base isbicarbonate, HCO3-. Because the hydrogen ion attaches and isnot free in solution, the pH will not change dramatically. The weakacid that forms will not dissociate into hydrogen ions toany great extent.What do you mean by pKa?
pKa is a measure of acid strength. It depends onthe identity and chemical properties of the acid. pH is a measureof [H+] in a solution. For acids, the smaller the pKa, themore acidic the substance is (the more easily a proton is lost,thus the lower the pH).How do you determine pKa?
To create a more manageable number, chemists define thepKa value as the negative logarithm of the Ka value:pKa = -log Ka. If you already know the pKa value foran acid and you need the Ka value, you find it by taking theantilog. In practice, this means raising both sides of the equalityto exponents of 10.What pKa is basic?
pKa Definition. pKa is the negativebase-10 logarithm of the acid dissociation constant(Ka) of a solution. The lower the pKa value,the stronger the acid. For example, the pKa of acetic acidis 4.8, while the pKa of lactic acid is 3.8.What happens when pKa equals pH?
Remember that when the pH is equal to thepKa value, the proportion of the conjugate base andconjugate acid are equal to each other. As the pHincreases, the proportion of conjugate base increases andpredominates. If the pH is at least 2.0 pH unitsbelow the pKa, then the conjugate acid is at least 99% ofthe total.Is a higher pKa more acidic?
A lower pKa means the Ka value is higherand a higher Ka value means the acid dissociatesmore readily because it has a larger concentration ofHydronium ions (H3O+). If you have a low pKb,then your Kb value is high.How do you go from pKa to pH?
The strength of an acid is measured by both itspH and its pKa, and the two are related by theHenderson-Hasslebalch equation. This equation is: pH =pKa + log[A-]/[AH], where [AH] is theconcentration of the acid and [A-] is the concentrationof its conjugate base after dissociation.What does the pKa of a drug mean?
pKa is a different term than pH pH is simply a measure of the [H+] concentration in agiven solution. pKa is defined as the pH were a drugexists as 50% ionized and 50% unionized. If pKa - pH = 0,then 50% of drug is ionized and 50% isunionized.Is pKa the same as pH?
The pKa is a measure of the strength of an acid.Specifically, it's the negative log of the dissociation constantfor an acid in water. There is no way to convert pKa topH because they're not equivalent. The pH is ameasure of acid in solution.Is nh2 an acid or base?
Ammonia is actually itself a weak base, so itsconjugate base NH2- is an incredibly strong base soit can get an extra proton to regenerate NH3 which is much morestable. As a result of the relative basicity of NH2- andCl-, we can determine that acid chlorides are much morereactive than amides.Is a high pKa a strong acid?
Remember from general chemistry that pKa is equalto the negative log of the Ka. And the lower the pKa valuethe stronger the acid. And from general chemistry,the stronger the acid the weaker the conjugate base.Because for an acid to be strong the conjugate basemust be weak to resist reprotonation.Is HCl an acid or base?
On the other hand, substances such as hydrochloricacid, HCl, are held together by polar ionic bonds andwhen placed into water the hydrogen will break away to formhydrogen ions, making the liquid acidic. HCltherefore has a very low pH and is a very strong acid. Weakacids, with pH 5 or 6 are slightly morecomplex.