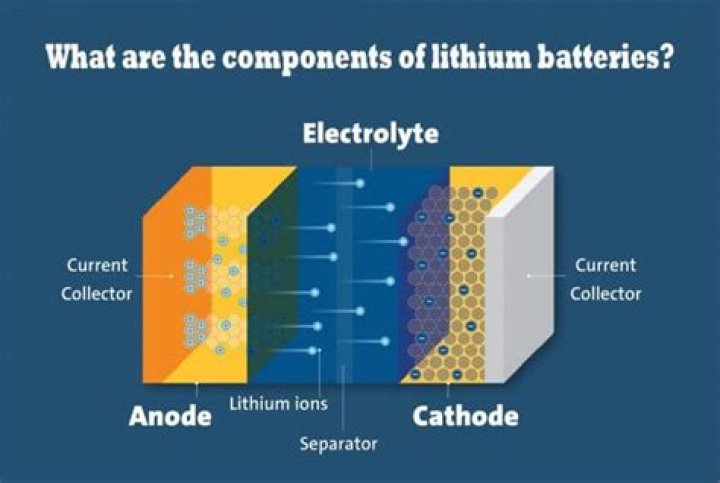
What is electrolyte in battery?
.
Then, what is the electrolyte in a battery made of?
These batteries typically contain a carbon anode, a cathode made of lithium cobalt dioxide and an electrolyte containing a lithium salt in an organic solvent.
Furthermore, what is the cathode of a battery? A cathode is a terminal through which electric current flows out of a polarized electrical gadget, wherein the direction of electric current is opposite to the direction of the flow of the electron. In this manner, electrons flow around the cathode terminal while current flows far from it.
Thereof, what is the best electrolyte for a battery?
In the lead acid battery, sulfuric acid and water are the electrolyte. It also supplies the sulfate ions required for the liberation of oxygen molecules into solution. Fill a beaker half full of water. For an electrolyte solution, distilled water is the best choice.
How do you measure electrolytes in a battery?
A hydrometer is used to measure the specific gravity of the electrolyte solution in each cell. It's a tool used to measure the density or weight of a liquid compared to the density of an equal amount of water. A lead acid battery cell is fully charged with a specific gravity of 1.265 at 80° F.
Related Question AnswersCan you put vinegar in a car battery?
Remove the damaged battery and put it in a plastic bag. Wipe away any white, solid materials (loose corrosion) using a cloth. Pour some white vinegar into a cup or a bowl. Dip a cotton swab into the white vinegar and then use it to wipe the contacts.How do you replace an electrolyte battery?
How to Change Electrolytes in a Lead-Acid Battery- Plug the battery charger into a wall electrical outlet and turn on the charger; this will break up any lead sulfate crystals that have formed on the battery plates.
- Allow the battery to cool down for at least one hour after charging.
- Loosen and remove the battery vent caps using the vent wrench.
How do you discharge a battery?
Specifically, if you want to fully discharge a typical car battery (12V, 60 A hr), all you need is a 20 ohm, 10 W resistor (or equivalent), and connect it across the battery terminals. Leave it connected for about 4 days, and with a voltmeter verify that the voltage is zero.How do you make an electrolyte battery?
In the lead acid battery, sulfuric acid and water are the electrolyte. It also supplies the sulfate ions required for the liberation of oxygen molecules into solution. Fill a beaker half full of water. For an electrolyte solution, distilled water is the best choice.What makes a good battery electrolyte?
In the lead acid battery, sulfuric acid and water are the electrolyte. For an electrolyte solution, distilled water is the best choice.How much electrolyte is in a battery?
Deep-cycle batteries use a dense electrolyte with an SG of up to 1.330 to achieve high specific energy, starter batteries contain an average SG of about 1.265 and stationary batteries come with a low SG of roughly 1.225 to moderate corrosion and promote longevity.What type of energy is in a battery?
chemical energyCan you put water in a battery instead of acid?
In the old days, most batteries were not sealed and they are empty when new. The acid is only added after you purchased the battery. Once they are in service you only add distilled water in them. Adding battery acid instead of distilled water will reduce the capacity of the battery.Can you add electrolyte to a battery?
The only electrolyte that can be used in a lead-acid battery is sulfuric acid. Adding anything but water to a battery can instantly damage it, but some substances are worse than others. For example, baking soda can neutralize the sulfuric acid present in a battery's electrolyte solution.Can I put new acid in my battery?
First the safe answer, no never add acid to a battery, just distilled water. Older batteries can suffer from sulfation, a buildup of lead sulfate on the plates of the battery inside. This can effectively weaken the acid to water ratio.Can you put aspirin in a battery?
Put two tablets of aspirin in each battery cell and wait no more than 1 hour (the acetylsalicylic acid combines with the sulfuric acid to get off one more charge.) Be warned that adding aspirin will shorten the battery life, as the aspirin will react with the sulfuric acid to form acetic acid.Can you put baking soda in a battery?
When the battery cells are all empty place your battery to one side and take ½ lb of your baking soda and add it to your bucket of old battery acid. This will neutralize the battery acid so you can safely dispose of it later.What happens when you put salt water in a battery?
So what happens if you put a battery in water? Salt dissolves in water to leave behind positively-charged sodium ions and negatively-charged chlorine ions. Once you put a battery in the water, the sodium ions migrate toward the “negative tank” and the chlorine ions migrate toward the “positive tank”.How do I bring my battery back to life?
Bring Dead Lead Acid Battery to Live Again- Step 1: Preparing the Battery. 3 More Images.
- Step 2: Fill the Water Inside the Battery.
- Step 3: Mix Water With Acid and Charging.
- now pull the wasted water from the top of 3 holes by the syringe and let the battery charging.
- 39 Discussions.
How do you restore a lead acid battery?
How to Revive Old Lead Acid Batteries- Step 1: You Will Need. -Multimeter.
- Step 2: Measure Battery Voltage. Connect multimeter to your battery and check voltage.
- Step 3: Inspect the Cells. Place paper towels on your working areas.
- Step 4: Add Water.
- Step 5: Turn on Your PSU.
- Step 6: Testing Battery.
- Step 7: Edit: 28.04.