Coulomb's law is a function developed in the1780s by physicist Charles-Augustin de Coulomb. It explainshow strong the force will be between two electrostatic charges.Electrostatic means electric charges without anymotion..
Thereof, how do you explain Coulomb's law?
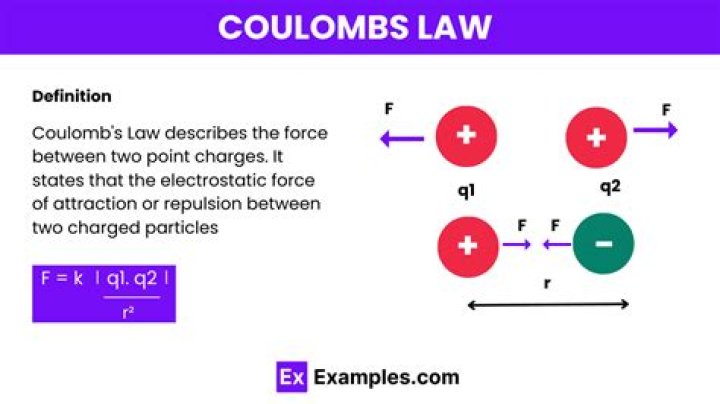
Coulomb's law states that: The magnitude of theelectrostatic force of attraction or repulsion between two pointcharges is directly proportional to the product of the magnitudesof charges and inversely proportional to the square of the distancebetween them. The force is along the straight line joiningthem.
One may also ask, why is Coulomb's law important? It signifies, the inverse square dependence of electricforce. It can also be used to provide relatively simple derivationsof Gauss' law for general cases accurately. Finally, thevector form of Coulomb's law is important as it helpsus specify the direction of electric fields due tocharges.
Also asked, what is Coulomb's law for dummies?
Coulomb's Law is one of the basic ideas ofelectricity in physics. The law looks at the forces createdbetween two charged objects. As distance increases, the forces andelectric fields decrease. Similar charges such as two positive ortwo negative push away from each other.
How does Coulomb's law apply to situations?
Coulomb's law allows us to find the force betweentwo point charges. "There will be zero net electric force on thecharge in the middle due to the other charges. Using coulomb'slaw, the force due to the +Q charge is positive, and the forcedue to the −Q charge is negative.
Related Question Answers
What is q1 and q2 in Coulomb law?
Coulomb's law establishes the force that a“charged particle”, say q1, experiences becauseof another charged particle, say q2, around it. The force isproportional to the product of charges and inversely proportionalto the square of the distance between them. The formula can then bewritten as -What is the SI unit of Coulomb law?
The coulomb (symbolized C) is the standardunit of electric charge in the International System ofUnits (SI). It is a dimensionless quantity, sharingthis aspect with the mole. A quantity of 1 C is equal toapproximately 6.24 x 1018, or 6.24quintillion.What are the three laws of electrostatics?
- First law: Like charges of electricity repel each other,whereas unlike charges attract each other.
- Second law:According to this law, the force exerted between twopoint charges. i) is directly proportional to the product of theirstrengths. ii) is inversely proportional to the square of thedistance between them. and.
Is Coulomb's law a universal law?
Coulomb's law only works for a point charge, andnot for all charge distributions. As it only works in certainsituations (as does Ohm's Law), then it is notuniversal. A universal law will work in anysituations.How do you find coulombs?
How to Calculate Coulombs - charge (coulomb, C) = current (ampere, A) × time (second,s).
- energy transformed (joule, J) = potential difference (volt, V)× charge (coulomb, C).
- F = kq1q2 ÷ r2.
What charge means?
An electric charge is the property of matterwhere it has more or fewer electrons than protons in its atoms.Electrons carry a negative charge and protons carry apositive charge. Matter is positively charged if itcontains more protons than electrons, and negatively chargedif it contains more electrons than protons.How did Coulomb measure charge?
June 1785: Coulomb Measures the Electric Force.Charles Augustin Coulomb (top) used a calibrated torsionbalance (bottom) to measure the force between electriccharges. Twenty-three centuries later, Benjamin Franklinattributed this effect to the existence of two electrical fluids,one positive and the other negative.Where does Coulomb's law come from?
Formulated by the 18th-century French physicistCharles-Augustin de Coulomb, it is analogous to IsaacNewton's law of gravity. Both gravitational and electricforces decrease with the square of the distance between theobjects, and both forces act along a line betweenthem.What is the formula of Coulomb law?
Coulomb's Law Equation where Q1 represents the quantity of chargeon object 1 (in Coulombs), Q2 represents thequantity of charge on object 2 (in Coulombs), and drepresents the distance of separation between the two objects (inmeters). The symbol k is a proportionality constant known as theCoulomb's law constant.How do you find the charge of an object?
To determine the charge on an object,determine the number of excess protons or excess electrons.Multiply the excess by the charge of an electron or thecharge of a proton - 1.6 x 10-19 C.Finally, adjust the sign of the object to + or -.7.How do objects become charged?
An object gets charged when it's atomslose or gain an electron to become an ion. For example: AHydrogen atom has one positive proton and one negative electron.The fur is positively charged because it lost electrons andthe rod is negatively charged because it gainedelectrons.What is Coulomb law in vector form?
Coulomb measured the force between twopoint charges and found that it varied inversly. as the square ofthe distance between the charges and was directly proportional tothe product of the. magnitude of two charges and acted along theline of joining the two charges.What if Coulomb's force disappears?
If the Coulomb force disappears on Earth ,will it result in a supernova explosion? Disappearance ofcoulomb force removes the energy barrier that holds nucleiof the atoms apart. Disappearance of coulomb force removesthe energy barrier that holds nuclei of the atomsapart.Who discovered Coulomb's law?
Charles-Augustin de Coulomb
What are the limitations of Coulomb's law?
Limitations of Coulomb's Law: Its only applicablefor point charges at rest. Its only applicable in dose cases whereinverse square law is obeyed. It is difficult to apply theCoulomb's law when the charges are in arbitraryshape.How does Coulomb's law relate to ionization energy?
Ionization Energies of the Atoms. In order toremove an electron from an atom, we have to raise the potentialenergy from its negative value to zero. According toCoulomb's law, we expect electrons closer to the nucleus tohave a lower potential energy and thus to require moreenergy to remove from the atom.What is Coulomb's constant equal to?
Coulomb's constant. Coulomb's constant,the electric force constant, or the electrostaticconstant (denoted ke ) is a proportionality constantin equations relating electric variables and is equal to ke= 8.9875517873681764×10 9 N. · m2/C 2 (i.e. m/F).Why do like charges repel each other?
Like charges repel, unlike chargesattract Two electrons will tend to repel each otherbecause both have a negative electrical charge. Two protonswill also tend to repel each other because they both have apositive charge.What current flows in one direction?
There are two main types of current in our world.One is direct current (DC) which is a constant streamof charges in one direction. The other is alternatingcurrent (AC) that is a stream of charges that reversesdirection.