A protecting group or protective group is introduced into a molecule by chemical modification of a functional group to obtain chemoselectivity in a subsequent chemical reaction. It plays an important role in multistep organic synthesis. The acetal is then called a protecting group for the carbonyl..
In this regard, what makes a good protecting group?
Protecting groups are used in synthesis to temporarily mask the characteristic chemistry of a functional group because it interferes with another reaction. A good protecting group should be easy to put on, easy to remove and in high yielding reactions, and inert to the conditions of the reaction required.
Secondly, what is a silyl group? Silyl ethers are a group of chemical compounds which contain a silicon atom covalently bonded to an alkoxy group. Silyl ethers are usually used as protecting groups for alcohols in organic synthesis.
Likewise, people ask, what are blocking groups?
Blocking Group Definition: Blocking groups, in aromatic substitution reactions, is the functional group that can be readily installed and uninstalled.
How do you Deprotect alcohol?
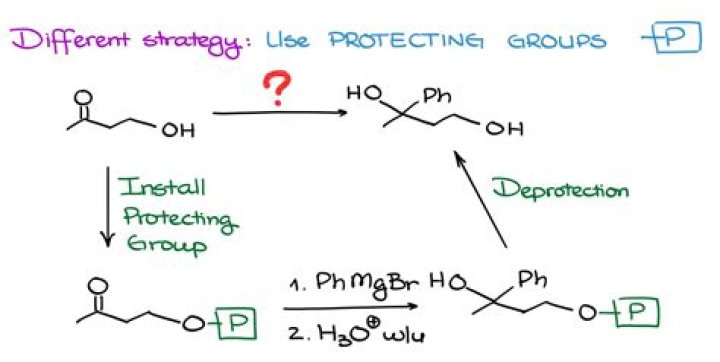
Example
- The silyl ether protecting group can be removed by reaction with an aqueous acid or the fluoride ion.
- By utilizing a protecting group a Grignad reagent can be formed and reacted on a halo alcohol. 1) Protect the Alcohol.
- 2) Form the Grignard Reagent.
- 3) Perform the Grignard Reaction.
- 4) Deprotection.
Related Question Answers
How do I remove BOC protecting group?
Protection of the amine can also be accomplished in acetonitrile solution using 4-dimethylaminopyridine (DMAP) as the base. Removal of the BOC in amino acids can be accomplished with strong acids such as trifluoroacetic acid in dichloromethane, or with HCl in methanol.How can we protect carboxylic acids?
Carboxylic acid protecting groups Protection of carboxylic acids: Methyl esters – Removed by acid or base. Benzyl esters – Removed by hydrogenolysis. tert-Butyl esters – Removed by acid, base and some reductants.What does Tbaf?
As a source of fluoride ion in organic solvents, TBAF is used to remove silyl ether protecting groups. It is also used as a phase transfer catalyst and as a mild base. As a deprotecting group, TBAF in DMSO will convert O-silylated enolates into carbonyls.What are Ketals?
In organic chemistry, a ketal is a functional group derived from a ketone by replacement of the carbonyl (C=O) group by two alkoxy groups. The IUPAC once declared the term "ketal" obsolete, but later accepted it as a subclass of acetals. Therefore, a ketal can also be defined as "an acetal derived from a ketone."How would you remove the trityl protecting group from an alcohol?
A trityl group is removed by strong acid hydrolysis as is benzenesulfonyl. The most useful N-protecting group would appear to be alkoxymethyl (Section 4.07. 1.3.What is Chemoselective reaction?
Chemoselective: A reaction that operates exclusively on one functional group in the presence of other functional groups. This NaBH4 reduction is chemoselective because the ketone is reduced but the ester is unchanged.What is the purpose of a silyl ether?
What is the purpose of a silyl ether? A) To protect esters from organometallic reagents and other reagents. B) To protect ketones from organometallic reagents and other reagents. C) To protect alcohols from organometallic reagents and other reagents. D) To prevent the formation of carboxylic acids.How can we protect ketone groups?
To protect a ketone using a straight chain alcohol, like methanol, you react two molecules of alcohol and a ketone and produce one molecule of water and an acetal. To protect a ketone using a diol, you react one molecule of diol and a ketone and produce one molecule of water and an acetal.How does Harvard housing work?
How does the housing system at Harvard work? Incoming freshman get matched to roommates and freshman dorms based on a survey they fill out, then after that they form groups of up to 8 people and each group gets randomly assigned to one of the 12 upperclassmen houses.Do Grignards react with alcohols?
Grignard Reaction With Alcohols If there are any alcohols on a carbon chain, then the Grignard reagent will react with the hydrogen on the alcohol before it will react with the carbonyl carbon. The same thing occurs if water is added to the reaction before the Grignard reagent has reacted with the aldehyde/ketone.What is the functional group of ketone?
In chemistry, a ketone /ˈkiːto?n/ is a functional group with the structure RC(=O)R', where R and R' can be a variety of carbon-containing substituents. Ketones and aldehydes are simple compounds that contain a carbonyl group (a carbon-oxygen double bond).What makes an acetal a unique functional group?
An acetal is a functional group with the connectivity R2C(OR')2). Acetals are formed from and convertible to aldehydes or ketones and have the same oxidation state at the central carbon, but have substantially different chemical stability and reactivity as compared to the analogous carbonyl compounds.What can Grignard reagents react with?
Grignard reagents are formed by the reaction of magnesium metal with alkyl or alkenyl halides. They're extremely good nucleophiles, reacting with electrophiles such as carbonyl compounds (aldehydes, ketones, esters, carbon dioxide, etc) and epoxides. Similar to or the same as: very similar to organolithium reagents.What do acetals protect?
Cyclic acetals offer stability against all types of nucleophiles and bases. Deprotection is often performed by acid catalyzed transacetalization in acetone or hydrolysis in wet solvents or in aqueous acid. Some strong oxidation agents may cleave acetals such as HClO4 in CH2Cl2.What is acetal linkage?
An acetal is a functional group formed when an aldehyde reacts with an alcohol to form a functional group with two ether groups on the same carbon. A glycosidic linkage is the bond that forms when the hemiacetal OH of the anomeric carbon reacts with another alcohol group to form an acetal.What is silylation reaction?
Silylation is the introduction of a (usually) substituted silyl group (R3Si) to a molecule. The process is the basis of organosilicon chemistry.