What does Peracid do in a reaction?
What does Peracid do in a reaction?
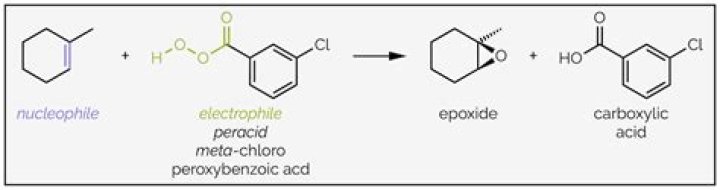
Reaction principle During the epoxidation of alkenes, an oxygen atom is transferred from the peracid to the C=C double bond thus forming an oxirane ring. Since the transferred oxygen atom carries a positive charge, peracids must be considered electrophilic oxidizing agents.
What does peroxy acid do?
The peroxy acids are used primarily as oxidizing agents; they readily add oxygen to alkenes to give epoxides and are used to convert ketones to esters and amines to nitro compounds, amine oxides, or nitroso compounds.
What type of oxidizing agent is used in a Baeyer Villiger Oxidation?
hydrogen peroxide
This reaction can be accomplished using hydrogen peroxide, 3-chloroperbenzoic acid (m-chloroperoxybenzoic acid), peroxyacetic acid, or peroxytrifluoroacetic acid as the oxidizing agent. This reaction is also useful in the synthesis of lactones (cyclic esters) from cyclic ketones.
What is bare Villiger Oxidation?
The Baeyer-Villiger Oxidation is the oxidative cleavage of a carbon-carbon bond adjacent to a carbonyl, which converts ketones to esters and cyclic ketones to lactones. The Baeyer-Villiger can be carried out with peracids, such as MCBPA, or with hydrogen peroxide and a Lewis acid.
What is the meaning of Peracids?
peracid. / (pɜːrˈæsɪd) / noun. an acid, such as perchloric acid, in which the element forming the acid radical exhibits its highest valency. (not in technical usage) an acid, such as persulphuric acid, that contains the -OOH groupRecommended names: peroxo acid, peroxy acid.
What is Peracid formula?
Peracetic acid (also known as peroxyacetic acid, or PAA), is an organic compound with the formula CH3CO3H. This organic peroxide is a colorless liquid with a characteristic acrid odor reminiscent of acetic acid.
Why are Peroxy weaker than carboxylic acids?
Properties and uses. In terms of acidity, peroxycarboxylic acids are about 1000 times weaker than the parent carboxylic acid, due to the absence of resonance stabilization of the anion.
What is the given reaction known as Baeyer Villiger?
The given reaction is known as Baeyer – villiger oxidation. In this reaction, acetone is heated with perbenzoic acid to form methyl acetate. When an unsymmetrical aliphatic ketone is used, the oxygen atom is inserted between carbonyl carbon and the larger alkyl group.
Is Baeyer Villiger reaction an oxidation or rearrangement reaction?
The Baeyer-Villiger rearrangement is the conversion of a ketone to an ester via the insertion of an oxygen atom next to the carbonyl. The reaction involves initial addition of a peroxide to the carbonyl carbon. The resulting adduct undergoes rearrangement to form the ester.
Which of the following is an example of Baeyer Villiger rearrangement?
1]hept-2-yl)ethanone is rearranged to an ester, bicyclo[2.2. 1]hept-2-yl acetate upon Baeyer villiger oxidation with mCPBA in dichloromethane and the subsequent hydrolysis of the ester gives the desired alcohol, bicyclo[2.2. 1]heptan-2-ol . It is another example of regioselectivity and stereospecificity.
Can I use hydrogen peroxide?
The Food and Drug Administration (FDA) classifies hydrogen peroxide as “generally recognized as safe” (GRAS) for humans at low doses. But the FDA warns that getting hydrogen peroxide on your skin may cause irritation, burning, and blistering.
How do you make performic acid?
Performic acid (PFA), an unstable colorless liquid, can be produced by reacting formic acid with hydrogen peroxide (H2O2).