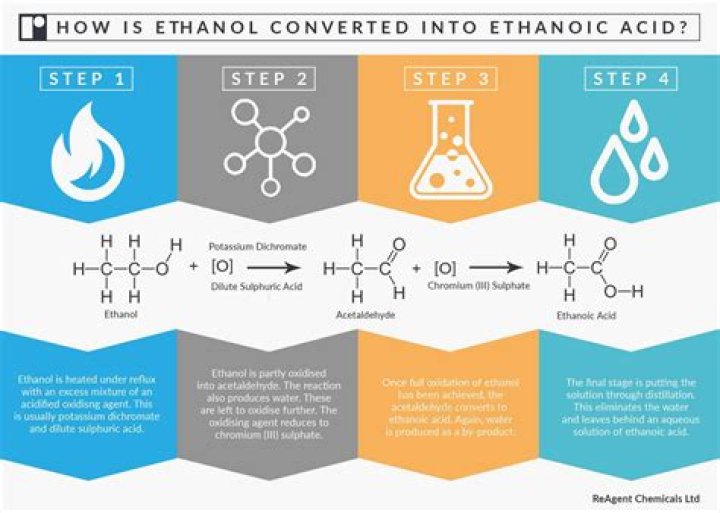
Nital is a solution of nitric acid and alcohol commonly used for etching of metals. Mixtures of ethanol and nitric acid are potentially explosive. This commonly occurs by gas evolution, although ethyl nitrate can also be formed. Methanol is not liable to explosion but it is toxic..
Similarly, what can you not mix with nitric acid?
Incompatibilities of concentrated nitric acid:
- Never mix concentrated nitric acid and organics, such as acetone, unless you are following a respectable procedure and use a blast shield and proper precautions.
- Never store mixtures of concentrated acids, particularly.
- concentrated acids by slow addition to ice or water in an.
Furthermore, can nitric acid explode? Contact between a broken organic solvent bottle and a container of nitric acid can lead to the release of nitrous fumes and a potential violent explosion. Explosions have also occurred when the organic acid is added to the nitric acid instead of the reverse.
Also Know, how do you make a 3% nital solution?
To make 100 ml of 3% nital solution, measure 97 ml of methanol into a measuring beaker and pour into a plastic acid-resistant lab bottle.
Does red fuming nitric acid react with water?
Red fuming nitric acid (RFNA) is a storable oxidizer used as a rocket propellant. It consists of 84% nitric acid (HNO3), 13% dinitrogen tetroxide and 1–2% water. RFNA increases the flammability of combustible materials and is highly exothermic when reacting with water.
Related Question Answers
Will baking soda neutralize nitric acid?
Store nitric acid in the original container. Dilute solutions must be stored in acid-resistant bottles. If less than 10ml of concentrated acid or 100ml of dilute acid is spilled, the spill can be neutralized by gently adding soda ash or sodium bicarbonate and rinsing with copious quantities of water.What happens when you mix nitric acid and hydrogen peroxide?
Concentrated nitric acid is also used to passivate equipment for use with hydrogen peroxide. In particular a number of researchers (1,2,3) have reported that at nitric acid concentrations of greater than 35%, a reaction occurs which may be violent in nature and which can produce heat and large volumes of NOx gas.Does nitric acid dissolve plastic?
Most plastics are resistant to dilute nitric acid.What can nitric acid dissolve?
Nitric acid is the primary choice here on several grounds. It is an oxidizing acid that can dissolve most metals to form soluble metal nitrates. It has poor oxidizing strength below 2 M but is a powerful oxidizing acid in concentrated form.How do you dispose of nitric acid?
Small amounts, under 300 ml, of dilute nitric acid can be disposed of down sinks or toilets as long as there is a large quantity of water used to further dilute the acid. Stop-up your sink using your stopper. Pour a gallon of water in the sink. Pour your dilute solution in the water and release the stopper.Why is nitric acid stored separately?
Concentrated nitric acid must be stored in a separate liquid-tight compartment within an acid cabinet. If nitric acid is mixed with a flammable organic compound, such as acetic acid, the heat from the oxidation and neutralization reactions is enough to ignite the flammable material.What does nitric acid react with?
Dilute nitric acid behaves as a typical acid in its reaction with most metals. Some precious metals, such as pure gold and platinum-group metals do not react with nitric acid, though pure gold does react with aqua regia, a mixture of concentrated nitric acid and hydrochloric acid.Can you make nitric acid?
Chemically, nitric acid is made by bubbling nitrogen dioxide into water. This can be done by reacting hydrochloric acid, a nitrate salt and copper. Around 80-grams of sodium nitrate, over 30-grams of copper and 100-mL of hydrochloric acid are the quantities needed.What happens when you mix nitric acid and alcohol?
Mixtures of ethanol and nitric acid are potentially explosive. This commonly occurs by gas evolution, although ethyl nitrate can also be formed. A solution of ethanol and nitric acid will become explosive if the concentration of nitric acid reaches over 10% (by weight).How do you make 2 Nital?
Nital is easily made by simply mixing nitric acid and methanol in the desired proportions, typically 1-10 % by volume nitric acid into methanol depending on whether a microetch or a macroetch is desired. Remember, alway add acid to the methanol (or water, as the case may be), and never the other way around.How do you make methanol?
About Methanol Methanol – CH3OH – is four parts hydrogen, one part oxygen and one part carbon. On an industrial scale, methanol is predominantly produced from natural gas by reforming the gas with steam and then converting and distilling the resulting synthesized gas mixture to create pure methanol.What is Nital used for?
Nital is a solution of nitric acid and alcohol commonly used for etching of metals. It is especially suitable for revealing the microstructure of carbon steels. The alcohol can be methanol, ethanol or methylated spirits. Mixtures of ethanol and nitric acid are potentially explosive.How does Nital etch work?
Nital Etch inspection works by exposing the material or part in question to a mixture of alcohol and nitric acid. Damaged areas will be darker than their surrounding areas.What is Nital etch inspection?
Nital Solution is a mixture of nitric acid and alcohol commonly used for etching steels. Nital Etch Test is a procedure for checking machining damage or grinding burn of a hardened steel component. It's also known as the Temper etch test, Nital inspection test, Nital NDT, MIL-STD-867C, AMS 2649C, etc.Does nitric acid react with water?
The nitrogen dioxide is absorbed in water to form nitric acid. The resulting acid-in-water solution (about 50–70 percent by weight acid) can be dehydrated by distillation with sulfuric acid. Nitric acid decomposes into water, nitrogen dioxide, and oxygen, forming a brownish yellow solution.What metals does nitric acid dissolve?
Although chromium (Cr), iron (Fe), and aluminium (Al) readily dissolve in dilute nitric acid, the concentrated acid forms a metal-oxide layer that protects the bulk of the metal from further oxidation.How dangerous is nitric acid?
Health Hazards Associated with Nitric Acid Nitric acid is an extremely corrosive acid capable of causing severe chemical burns very rapidly. If nitric acid mists are inhaled, health risks include corrosion of mucous membranes, delayed pulmonary edema, and even death. Contact with eyes can cause permanent cornea damage.How do you store nitric acid?
Store nitric acid in a secure area away from incompatible materials such as organic compounds, metals, alcohols or moisture. Keep container tightly closed in a cool, dry, well-ventilated area.What is concentrated nitric acid?
Commercially available nitric acid is an azeotrope with water at a concentration of 68% HNO3. This solution has a boiling temperature of 120.5 °C at 1 atm. It is known as "concentrated nitric acid". Pure concentrated nitric acid is a colourless liquid at room temperature.