What cis double bond?
What cis double bond?
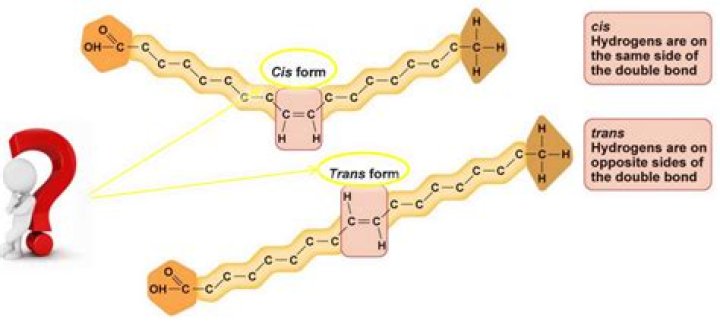
Unsaturated fatty acids have one or more double bonds. Each double bond may be in a cis or trans configuration. In the cis configuration, both hydrogens are on the same side of the hydrocarbon chain. In the trans configuration, the hydrogens are on opposite sides. A cis double bond causes a kink in the chain.
What is a cis bond?
This means that a molecule like 2-butene will have two possible isomers; the isomer where the two methyl groups are on the same side of the double bond is called “cis”, and the isomer where the two methyl groups are on the opposite side are called “trans”.
What structure is Transfat?
Trans fats are unsaturated fats with trans double bonds instead of cis bonds. The type of bond affects the shape of the fatty acid chain. A trans bond creates a straight chain, whereas a cis bond results in a chain that is bent. Trans fats may be monounsaturated or polyunsaturated.
What are cis fatty acids?
noun, plural: cis fatty acids. A fatty acid in a cis configuration, i.e. two hydrogen atoms adjacent to the double bond are on the same side of the chain. Supplement. Unsaturated fatty acids are those containing at least one double bond. This indicates that they can absorb additional hydrogen atoms.
What is cis addition?
An addition reaction of an alkene or an alkyne in which the net reaction is addition of two ligands to the multiple-bonded carbon atoms from the same face of the multiple bond is called a syn addition; one in which the net reaction is addition of the two ligands from the opposite faces of the multiple bond is called an …
What are cis molecules?
Cis–trans isomerism, also known as geometric isomerism or configurational isomerism, is a term used in organic chemistry. In the context of chemistry, cis indicates that the functional groups (substituents) are on the same side of some plane, while trans conveys that they are on opposing sides.
Is cis fat healthy?
Fatty acids with cis configuration are typical in natural foods. Most of the trans fatty acids are formed during the process of hydrogenation of vegetable oils. Trans fatty acids are not essential and have no known health benefit. In fact, similar to saturated fatty acids, they may increase the risk of heart diseases.
Why is cis fat better?
Health Consequences While consuming cis fats in unnaturally large quantities poses a health risk, unsaturated cis fats — monounsaturated and polyunsaturated fats — generally increase the levels of good cholesterol (HDL) in the body while also decreasing bad cholesterol.
What is threo and erythro?
Erythro and threo are two configurations in which molecules are written when the molecules have a chiral carbon atom. Erythro is the configuration when the same groups are on the same side of the carbon atom and Threo is the configuration when the same groups are on the opposite side of the carbon atom.
What is cis addition alkenes?
1,2-disubstituted alkenes are described as: cis- if the two alkyl groups, R-, are on the same side of the C=C. trans- if the two alkyl groups, R-, are on opposite sides of the C=C. these terms are inserted into the name as prefixes.
Why cis isomer is polar?
In cis isomer two polar groups attached to the same side of a restricted system and form an unsymmetrical molecule. Each bond have dipole movement and both the bond produce a resultant dipole moment in one direction, so cis form of alkene are polar.
Why is cis a fatty acid?
When the two hydrogen atoms stick out on the same side of the chain, the fatty acid is said to be in a cis configuration. This results in a kink due to the two hydrogen atoms repelling each other slightly. The more double bonds in the cis configuration, the less flexible is the fatty acid.



