What are the properties of liquids and solids?
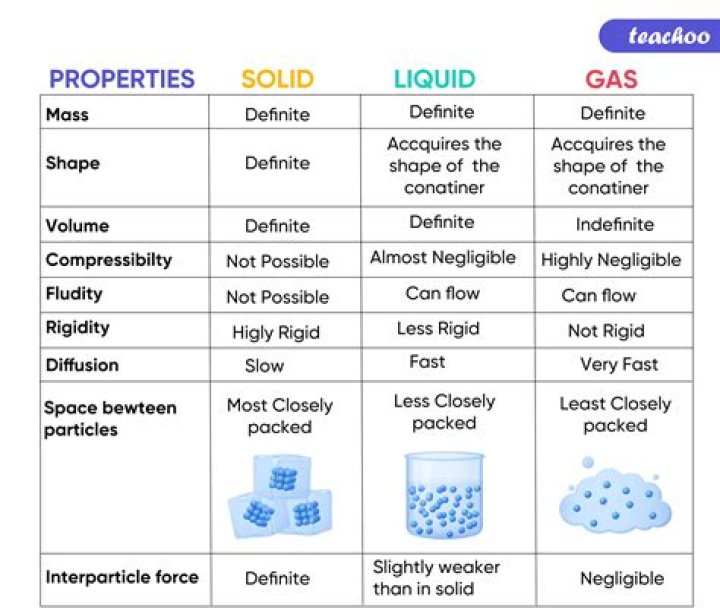
| Characteristic | Solid | Liquid |
|---|---|---|
| shape | definite | indefinite |
| volume | definite | definite |
| relative intermolecular interaction strength | strong | moderate |
| relative particle positions | in contact and fixed in place | in contact but not fixed |
.
Also question is, what are the properties of a liquid?
Liquids take on the shape of their container. The liquid state of matter is an intermediate phase between solid and gas. Like the particles of a solid, particles in a liquid are subject to intermolecular attraction; however, liquid particles have more space between them, so they are not fixed in position.
Also Know, what are the 4 properties of a solid? There are four different types of crystalline solids: molecular solids, network solids, ionic solids, and metallic solids. A solid's atomic-level structure and composition determine many of its macroscopic properties, including, for example, electrical and heat conductivity, density, and solubility.
Similarly one may ask, what are the 3 properties of a liquid?
The molecules does not have lot of space between them. The molecules can not squeezed closer to one another. Liquids have fixed volume but no fixed shape. They have fixed volume but they do not have fixed or definite shape.
What physical properties do solids and liquids have in common?
Sold, liquid, and gas all have volume and shape. They are all made up of atoms, molecules, or ions. Liquids and solids can be referred to as condensed phased because their particles are close together. Liquids and gases flow easily because their particles can move or slide past one another.
Related Question AnswersWhat are 4 properties of liquid?
A liquid is a nearly incompressible fluid that conforms to the shape of its container but retains a (nearly) constant volume independent of pressure. As such, it is one of the four fundamental states of matter (the others being solid, gas, and plasma), and is the only state with a definite volume but no fixed shape.What are 5 liquids?
Liquids can flow and assume the shape of their container.- Water.
- Milk.
- Blood.
- Urine.
- Gasoline.
- Mercury (an element)
- Bromine (an element)
- Wine.
What is the physical properties of liquids?
The most obvious physical properties of a liquid are its retention of volume and its conformation to the shape of its container. When a liquid substance is poured into a vessel, it takes the shape of the vessel, and, as long as the substance stays in the liquid state, it will remain inside the vessel.What are the six properties of liquids?
Liquids| Critical Temperature and Critical Pressure | Surface Tension |
|---|---|
| Viscosity | Hydrogen Bonding & the Anomalous Properties of Water |
What are the six properties of gases?
What Are Five Properties of Gases?- Low Density. Gases contain scattered molecules that are dispersed across a given volume and are therefore less dense than in their solid or liquid states.
- Indefinite Shape or Volume. Gases have no definite shape or volume.
- Compressibility and Expandability.
- Diffusivity.
- Pressure.
Is honey a liquid?
Honey is a supercooled liquid when stored below its melting point, as is normal. At very low temperatures, honey does not freeze solid; rather its viscosity increases. Like most viscous liquids, the honey becomes thick and sluggish with decreasing temperature.How many types of liquids are there?
Summary: There are two types of liquid water, according to research carried out by an international scientific collaboration. This new peculiarity adds to the growing list of strange phenomena in what we imagine is a simple substance.What liquids are compressible?
Compressibility of Liquids| Liquid | Compressibility, k | |
|---|---|---|
| Ethyl alcohol | 110 | 111 |
| Glycerine | 21 | 21 |
| Mercury | 3.7 | 3.8 |
| Water | 45.8 | 46.4 |
Is water a compressible fluid?
Water is slightly compressible. We often treat it as incompressible when doing fluid flow calculations because the pressure changes involved are too small to make an appreciable change to the density. The water at the bottom of the ocean is denser than at the surface due to the very large pressure there.What are examples of liquids?
Examples of liquids include:- Blood.
- Honey.
- Wine.
- Water.
- Mercury (a liquid metal)
- Oil.
- Milk.
- Acetone.



