What are some examples of endothermic?
- Photosynthesis.
- Melting ice.
- Evaporating liquid water.
- Sublimation of carbon dioxide (dry ice)
- Cracking of alkanes.
- Thermal decomposition reactions.
- Electrolytic decomposition of sodium chloride into sodium hydroxide and hydrogen chloride.
- Dissolving ammonium chloride in water.
.
Beside this, what are three examples of endothermic reactions?
These examples could be written as chemical reactions, but are more generally considered to be endothermic or heat-absorbing processes:
- Melting ice cubes.
- Melting solid salts.
- Evaporating liquid water.
- Converting frost to water vapor (melting, boiling, and evaporation, in general, are endothermic processes.
Furthermore, what are some examples of endothermic and exothermic reactions? Examples of Endothermic and Exothermic Processes
- Dissolving ammonium chloride in water.
- Cracking alkanes.
- Nucleosynthesis of elements heavier than nickel in stars.
- Evaporating liquid water.
- Melting ice.
Similarly, what are some examples of exothermic?
Some examples of exothermic processes are:
- Combustion of fuels such as wood, coal and oil petroleum.
- Thermite reaction.
- Reaction of alkali metals and other highly electropositive metals with water.
- Condensation of rain from water vapor.
- Mixing water and strong acids or strong bases.
- Mixing acids and bases.
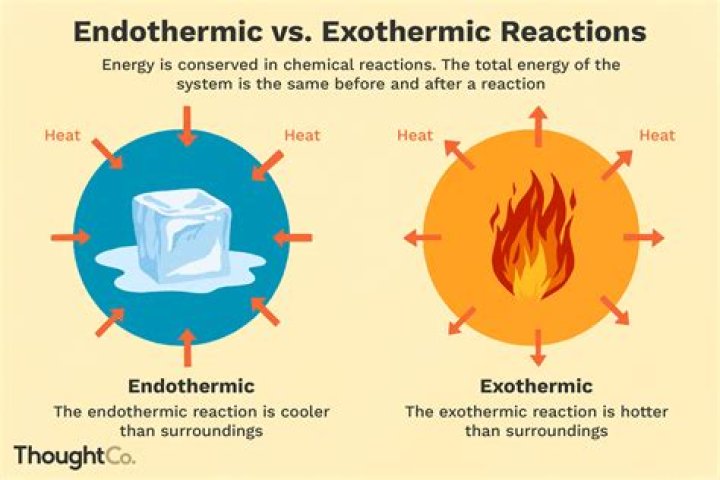
Which is an endothermic process?
An endothermic process is any process which requires or absorbs energy from its surroundings, usually in the form of heat. It may be a chemical process, such as dissolving ammonium nitrate in water, or a physical process, such as the melting of ice cubes.
Related Question AnswersIs endothermic hot or cold?
An endothermic reaction is when heat is needed by the reaction, so it draws heat from its surroundings, making them feel cold. Just like that ice pack.Is boiling water exothermic?
Because we must add heat, boiling water is a process that chemists call endothermic. Clearly, if some processes require heat, others must give off heat when they take place. It is less intuitive to grasp that when a gas condenses to a liquid, heat is given off and the process is exothermic.What do you mean by endothermic?
The definition of endothermic is a chemical reaction that is accompanied by the absorption of heat, or an organism that generates heat to maintain its temperature. A chemical reaction that works only if heat is absorbed is an example of a reaction that would be described as endothermic.Why is endothermic important?
An endothermic reaction is one that absorbs energy in the form of heat or light. Many endothermic reactions helps us i our daily life. You should also note that carbon dioxide is produced during this reaction. The chemical reaction that takes place when fuels burn has both positive and negative consequences.How do you know if its endothermic or exothermic?
If the energy level of the reactants is higher than the energy level of the products the reaction is exothermic (energy has been released during the reaction). If the energy level of the products is higher than the energy level of the reactants it is an endothermic reaction.What is the most endothermic reaction?
Once you are in space then ion engines are the latest trend though. The most endothermic reactions are entropy driven. In order to absorb heat from the environment, energy must be redistributed in an even less available form than random energy at that temperature.What are the uses of endothermic reactions?
- Everyday uses of exothermic reactions include self-heating cans and hand warmers.
- Everyday uses of endothermic reactions include instant ice packs which can be used to treat sports injuries.



