Is p-nitrophenol an acid or base?
Is p-nitrophenol an acid or base?
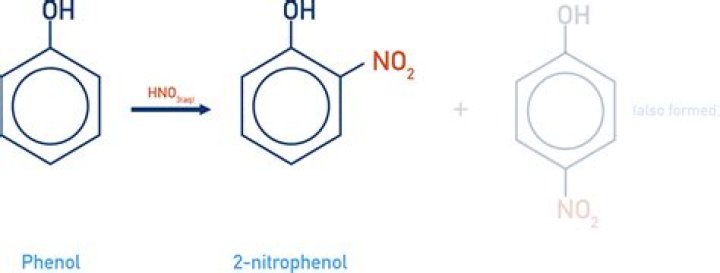
The compound p-nitrophenol is more acidic than just plain old phenol (see structures below). Why do you think this might be? The proton on the OH group of the p-nitrophenol compound is about 1000 times more likely to dissociate than the proton on the OH group of the phenol compound.
Which is more acidic para nitrophenol or?
p- Nitrophenol is a stronger acid than o-nitrophenol.
Why para nitrophenol is most acidic?
The decrease in electron density of the O – H bond of p-nitrophenol, the polarity of O – H bond decreases. The electron withdrawing group (-NO ), withdraws electrons and disperses the negative charge. Therefore, -NO group stabilizes the phenoxide ion. Hence p-nitrophenol is more acidic than phenol.
Is para nitrophenol more acidic than phenol?
Para-nitrophenol is more acidic than phenol due to the presence of an electron withdrawing group, -NO2 group, which enhances the acidic strength of the compound by stabilizing the phenoxide ion and the ortho- and para-positions in the benzene ring becomes electron rich.
Why is para nitrophenol yellow?
pH indicator The yellow color of the 4-nitrophenolate form (or 4-nitrophenoxide) is due to a maximum of absorbance at 405 nm (ε = 18.3 to 18.4 mM−1 cm−1 in strong alkali).
What is the structure of para nitrophenol?
C6H5NO3
4-Nitrophenol/Formula
Which is more acidic ortho or para?
Ortho nitrophenol is less acidic than para nitrophenol because of the intermolecular hydrogen bonding which makes the loss of proton very difficult. So, para nitrophenol is more acidic.
Why is para more acidic than meta?
In benzene compounds , meta position never contributes in the resonance. Due to resonance stabilization of the conjugate base formed para nitro phenol is more acidic than meta nitro phenol whose conjugate base is stabilised by inductive effect only.
Why ortho and para nitrophenol are more acidic than meta nitrophenol?
An inductive effect is an electronic effect due to the polarisation of σ bonds within a molecule or ion. Thus, ortho and para-nitrophenols are more acidic than m-nitrophenol is a little less acidic than p-nitrophenol because of intramolecular hydrogen bonding which makes the loss of a proton difficult to remove.
Why para nitrophenol have higher boiling point than Ortho nitrophenol?
p-Nitrophenol has intermolecular hydrogen bonding so it has more boiling point and less volatile than o-nitrophenol which has intramolecular hydrogen bonding.
Why is para more acidic than Ortho?
Is Ortho nitrophenol colorless?
ONPG, ortho-nitrophenyl-galactoside, is a colorless compound cleaved by β-galactosidase to yield the soluble compound, ortho-nitrophenol (ONP), which absorbs light at a maximum wavelength of 420 nm and is yellow in color.