Magnesium oxide is considered ionic. This means that the bonds that hold magnesium and oxygen together are formed between atoms that give electrons or.
Accordingly, is MgO ionic or covalent bond?
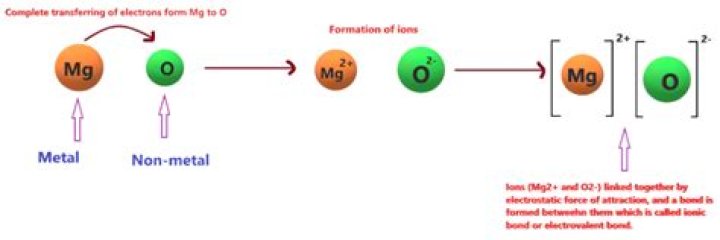
So 'O'-atom has a tendency to gain two electron ,so that it attain the octet rule . Hence by transfer of two electron from Mg to 'O'-atom , MgO is made of from Mg2+ and O-2 ion . So MgO is ionic compound . It is not a covalent compound.
Similarly, what type of bond exist in MgO? ionic bond
Also asked, is MgO an ionic compound?
Magnesium oxide
Is MgO polar or nonpolar or ionic?
Introduction:
| Compound | Type and bonding | Graphic Image |
| Oxygen O2 | non polar equal sharing of electrons | Graphic |
| Magnesium Oxide MgO | ionic transfer of electrons | Graphic |
| Water H2O | polar unequal sharing of electrons | Graphic |
| Sodium Fluoride NaF | ionic transfer of electrons | Graphic |
Related Question Answers
Is h2o a covalent bond?
A water molecule, abbreviated as H2O, is an example of a polar covalent bond. The electrons are unequally shared, with the oxygen atom spending more time with electrons than the hydrogen atoms. Since electrons spend more time with the oxygen atom, it carries a partial negative charge.Is HCL polar or nonpolar?
HCL is a polar molecule as chlorine has a higher electronegativity than the hydrogen. Thus, it attracts electrons to spend more time at its end, giving it a negative charge and hydrogen a positive charge. How do you know if Br2 is polar or nonpolar?Is NaCl a covalent bond?
Ionic bonds usually occur between metal and nonmetal ions. For example, sodium (Na), a metal, and chloride (Cl), a nonmetal, form an ionic bond to make NaCl. In a covalent bond, the atoms bond by sharing electrons. Covalent bonds usually occur between nonmetals.Is o2 a polar covalent bond?
Explanation: Diatomic oxygen is made up of the same two elements, and they equally share the 4 electrons that make up the double bond between them. They're equally electronegative, which means that there are not any partial charges for each element. Since neither atom pulls harder, it's a non-polar covalent bond.Is h2o covalent or ionic?
H2O, more commonly known as water, is a covalent compound. This type of compound is the result of atoms, usually from nonmetal elements, sharing electrons. Water has a special type of covalent bond called a polar covalent bond.Is co2 a covalent bond?
Carbon dioxide is covalent bond. Carbon cannot form ionic bond because in the simplest sense carbon can neither lose 4 electrons for stablisation (Due to it's strong attraction to the nucleus) nor gain 4 electrons (Since it's small nucleus with 6 protons cannot manage extra 4 electrons).What type of bond is NH4Cl?
There are two types of chemical bond present in NH4Cl. 1. Ionic bonding. Because the cation ammonium (NH4) has a single electron in its valence shell, and Chlorine needs a single electron to fill its valence shell, it takes that one, and they stick together.Is NaF a polar covalent bond?
Bonds with partially ionic and partially covalent character are called polar covalent bonds. Formation of Sodium FlourideThe attraction of oppositely charged atoms and the transfer of electrons leads to the formation of an ionic compound. In this case, NaF.Does magnesium and oxygen form ionic bonds?
Magnesium is a metal and oxygen is a nonmetal and that reaches the requirements to be an ionic bond. Hence magnesium oxide is ionic. If however the reaction involves two nonmetals forming a molecule then it is considered a covalent.What type of bond is CO?
Carbon monoxide consists of one carbon atom and one oxygen atom, connected by a triple bond that consists of twocovalent bonds as well as one dative covalent bond. It is the simplest oxocarbon and is isoelectronic with the cyanide anion, the nitrosonium cation and molecular nitrogen.How do you tell if a compound is ionic or covalent?
There is a couple different ways to determine if a bond is ionic or covalent. By definition, an ionic bond is between a metal and a nonmetal, and a covalent bond is between 2 nonmetals. So you usually just look at the periodic table and determine whether your compound is made of a metal/nonmetal or is just 2 nonmetals.What type of bond is h2o?
covalent bond
How does magnesium bond with oxygen?
Since oxygen has a higher electronegativity, it attracts the two valence electrons from the magnesium atom and these electrons are transferred from the magnesium atom to the oxygen atom. Magnesium loses two electrons to form ( ext{Mg}^{2+}), and oxygen gains two electrons to form ( ext{O}^{2-}).How do you describe an ionic compound?
Ionic compounds are compounds made up of ions. These ions are atoms that gain or lose electrons, giving them a net positive or negative charge. Metals tend to lose electrons, so they become cations and have a net positive charge. Nonmetals tend to gain electrons, forming anions that have a net negative charge.How MgO is formed?
- MgO is ionic compound formed by Mg2+ cation & O2- anion. - Magnesium has electronic configuration [Ne] 3s2 . - It loses two electrons to form stable octet of Mg2+ cation. - Oxygen has electronic configuration [He] 2s2 2p4 .Is oxygen stable before bonding?
Atoms like oxygen (O) will form two covalent bonds because they already have six valence electrons and only need two more electrons obtained by sharing. Hydrogen tends to form covalent bonds because a single covalent bond will fill its shell.Is AlCl3 ionic or covalent?
Fluorine (being very electronegative) will "steal" a lot of electron density from aluminium making the bonds more ionic in nature than covalent. But AlCl3 is covalent due to the low difference in electronegativity. Hence proved! Anhydrous AlCl3 is covalent in nature.What is meant by covalent bond?
A covalent bond, also called a molecular bond, is a chemical bond that involves the sharing of electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs, and the stable balance of attractive and repulsive forces between atoms, when they share electrons, is known as covalent bonding.Is CH bond polar?
Carbon–hydrogen bond. Using Pauling's scale—C (2.55) and H (2.2)—the electronegativity difference between these two atoms is 0.35. Because of this small difference in electronegativities, the C−H bond is generally regarded as being non-polar. In structural formulas of molecules, the hydrogen atoms are often omitted.