Is hydrogen carbonate ion acidic or basic?
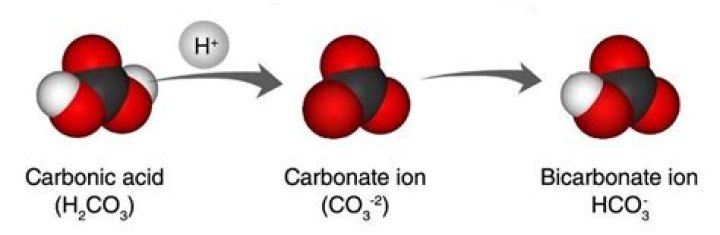
Is hydrogen carbonate ion acidic or basic?
CaCO3 + 2HCl → CaCl2 + CO2 + H2O The hydrogen carbonate anion is also a base. HCO3− + H3O+ → H2CO3 + H2O → CO2 + 2H2O It is actually stronger as a base than it is as an acid. Because of this, aqueous solutions of salts of hydrogen carbonates are weakly alkaline (basic) and are also active ingredients in many antacids.
What does hydrogen carbonate indicator detect?
Hydrogencarbonate indicator can detect increases and decreases in carbon dioxide concentration. It is normally red. However, an increase in carbon dioxide changes the indicator to yellow and a decrease in carbon dioxide changes it to purple. Photosynthesis only happens in the light.
Is hydrogen carbonate acidic or basic or amphoteric?
Yes, the bicarbonate ion is amphoteric.
Is sodium hydrogen carbonate an acid or base?
We know that sodium hydrogen carbonate is a basic salt because its parent base is strong (NaOH) and parent acid is weak (H2CO3).
Is hydrogen carbonate a base?
Bicarbonate, also known as hydrogen carbonate, is responsible for maintaining the balance of acids and bases in your body, i.e. the pH value. It is a base or alkaline, therefore an important “opponent” of acids. Bicarbonate works as an acid buffer.
Are hydrogen ions acidic?
High concentrations of hydrogen ions yield a low pH (acidic substances), whereas low levels of hydrogen ions result in a high pH (basic substances). It is neither acidic nor basic, and has a pH of 7.0. Anything below 7.0 (ranging from 0.0 to 6.9) is acidic, and anything above 7.0 (from 7.1 to 14.0) is alkaline.
How do you make a hydrogen carbonate indicator?
Can you give me a recipe for Bicarbonate (or Hydrogen carbonate) Indicator solution? Dissolve 0.2g of thymol blue and 0.1g creosol red in 20 cm3 ethanol. Weigh out 0.84g of sodium hydrogen carbonate (Analar) and dissolve this in about 900 cm3 of deionised or distilled water.
Which indicator is suitable for bicarbonate?
A bicarbonate indicator (hydrogencarbonate indicator) is a type of pH indicator that is sensitive enough to show a color change as the concentration of carbon dioxide gas in an aqueous solution increases. It is also used to test the carbon dioxide content during gaseous exchange of organisms.
Why the hydrogen carbonate ion is amphoteric?
Since they can donate a proton, all amphiprotic substances contain a hydrogen atom. Also, since they can act like an acid or a base, they are amphoteric.
Is hydrogen carbonate ion a strong base?
Acid-base chemistry The carbonate ion (CO32−) is a strong base. It is a conjugate base of the weakly acidic bicarbonate (IUPAC name hydrogen carbonate HCO3−), itself a strong conjugate base of the still weakly acidic carbonic acid. As such in aqueous solution, the carbonate ion seeks to reclaim hydrogen atoms.
Can nahco3 act as an acid?
Baking soda (NaHCO3) is an amphoteric compound that means it can act as acid as well as the base. The pH value of baking soda is around 8 which means, it is mildly alkaline or base in nature, so when baking soda is dissolved in water, it produces a basic solution.
Did you check the pH of sodium hydrogen carbonate?
The number of the ph for sodium hydrogen carbonate will be 8.27.