Fractional Values for Oxidation States The oxidation states are usually in whole numbers, but in some cases, they are in fractional numbers. Consider Fe3O4. In most cases, atoms of the same element in a given compound have the same oxidation states, but each atom can have a different state than the other..
Beside this, is argon a atom?
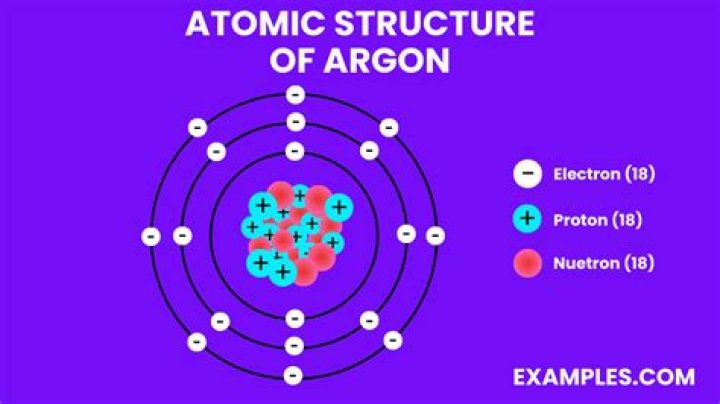
The argon atom has 18 electrons and 18 protons. Its outer shell is full with eight electrons. Under standard conditions argon is an odorless and colorless gas. It is also an inert gas, meaning that it typically doesn't react with other elements to form compounds.
Subsequently, question is, what is the charge for Argon?
| Name | Argon |
| Normal Phase | Gas |
| Family | Noble Gases |
| Period | 3 |
| Cost | 50 cents per 100 grams |
In this way, can Argon be an ion?
Argon Ion. Argon ions are then formed, and their amount is continuously increased by collisions of free electrons with neutral Ar atoms up to an equilibrium situation where a steady-state plasma is formed.
Is argon a metalloid?
Argon is a nonmetal — a noble gas.
Related Question Answers
Can argon gas kill you?
Argon is an inert gas, so isn't harmful in itself. However, it is an asphyxiant, which means it will, in sufficient concentration, displace the oxygen in the air, which will kill you. All you have to do is ensure that it can't concentrate.Is argon flammable?
Argon gas is colorless, odorless and non-flammable. It is non-toxic. The primary health hazard is asphyxiation by displacement of oxygen.Why is argon called the lazy one?
The name argon comes from the Greek word argos, "the lazy one." The name is based on argon's inability to react with anything. The discovery of argon created a problem for chemists. It was the first noble gas to be discovered.Do we need argon to live?
Whether you know it or not, you're breathing argon right now. But there's no need to be alarmed: This colorless, odorless gas makes up a mere 0.94 percent of the air around you, and it's so nonreactive that it has no effect on living organisms such as humans.Is Co a molecule?
Carbon Monoxide molecule. Carbon monoxide is a colorless, odorless, tasteless gas. A molecule of carbon monoxide (CO) contains one carbon atom and one oxygen atom.Why argon gas is used in spectrometer?
The Reasons of Using Argon Gas Isolate the atmosphere by flushing the surface so that the spectrometer won't detect the elements in the atmosphere. Spectrometer can detect the element, which has its characteristic peak below the wavelength 200nm correctly. In the composition of alloy, the element Argon is not ignored.Is argon heavier than air?
It's heavier than air and will tend to settle in low-lying areas. Argon is slightly water soluble. Argon is a member of a special group of gases known as the “noble” or “inert” gases. Other gases in this group are helium, neon and kryp- ton.Is oxygen a molecule?
Oxygen in the atmosphere is a molecule because it contains molecular bonds. It is not a compound because it is made from atoms of only one element - oxygen. This type of molecule is called a diatomic molecule, a molecule made from two atoms of the same type.Is argon gas an element or compound?
Argon is a noble gas, that consists 18 electrons and 18 protons. Its outershell is full with eight electrons. Under standard conditions it is an odorless and colorless gas. it is also an inert gas that it typically doesn't react with other element to form compounds.Is argon a covalent?
The extremely stable noble gasses, including helium, neon, argon, krypton, xenon and radon, are all also nonmetal covalent elements. These elements form bonds with one another by sharing electrons to form compounds.How many ions does argon have?
So for the element of ARGON, you already know that the atomic number tells you the number of electrons. That means there are 18 electrons in an argon atom. Looking at the picture, you can see there are two electrons in shell one, eight in shell two, and eight in shell three.What is the texture of argon?
Argon is a noble gas. It is colorless, odorless and extremely unreactive.What is the formula for Argon?
Argon Gas formula is discussed in this article. It is a noble gas with atomic number 18. It is monoatomic argon which is used in fluorescent tubes. The molecular formula of Argon Gas is Ar.Do noble gases form ions?
So, for noble gases, they already have their outermost shells filled completely so they need not donate or accept any electrons. For example: Argon has atomic no 18 ,i.e E.C = 2,8,8. They are already stable. So, they don't need to form ions.How many neutrons are in Argon?
22 neutrons
Why argon is used in plasma?
Argon prevents the oxidation of the surface as the plasma breaks the oxygen bond with the metal surface and carries it out of the chamber. Argon is also used after CF4 (or a CF4/O2 mixture) is used to etch a product (remove material).What type of bond does Argon form?
The third energy level will take the final 8 electrons and is full. Because all energy levels are full, Argon will not combine with other elements to form chemical bonds.Is argon positive or negative?
The flying charged particle continues on, leaving the knocked off electron behind in the electric field. Now, the argon atom has been separated into two pieces: an argon ion with positive charge, +1 unit, and the knocked-off electron with negative charge, -1 unit.What is Argon made of?
Argon is the third noble gas, in period 8, and it makes up about 1% of the Earth's atmosphere. Argon has approximately the same solubility as oxygen and it is 2.5 times as soluble in water as nitrogen . This chemically inert element is colorless and odorless in both its liquid and gaseous forms.