How many electrons does Ti 4 have?
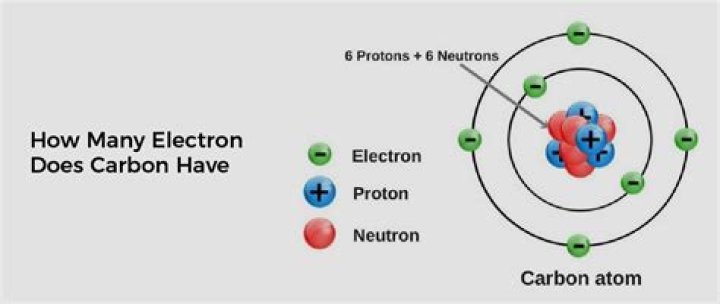
How many electrons does Ti 4 have?
22 electrons
For example, titanium can lose four electrons to form the titanium(IV) cation, Ti4+ . This ion has 22 protons and 18 electrons. So, to sum this up, an atom of titanium must have 22 electrons surrounding its nucleus.
What is the electronic configuration of 22ti?
Fact box
| Group | 4 | 1670°C, 3038°F, 1943 K |
|---|---|---|
| Block | d | 4.506 |
| Atomic number | 22 | 47.867 |
| State at 20°C | Solid | 48Ti |
| Electron configuration | [Ar] 3d24s2 | 7440-32-6 |
What element has the electron configuration 1s22s22p63s23p64s23d8?
Answer and Explanation: The atomic number is 28 which is of nickel (Ni). Hence, option b is the correct choice.
Why is Ti4 +( aq Colourless?
ions are colorless, unless a colored anion is present in the compound. The transition metal ions which have completely filled d-orbitals are colorless, as there are no vacant d-orbitals to permit promotion of the electrons. The transition metal ions which have completely empty d-orbitals are also colorless, Thus,Ti4+.
How many neutrons TI have?
Titanium has an atomic number of 22 so has 22 protons as well as 22 electrons. The mass number of Titanium is 48, which is the total number of protons and neutrons so, 48 – 22 = 26 = number of neutrons.
What is this element 1s22s22p63s2?
The electron configuration 1s22s22p63s23p2 is the element Silicon.
What is the element with the electron configuration 1s22s22p63s1?
Terms in this set (28) The electron configuration for Na+ is 1s22s22p6. When determining the electron configuration of an ion, first write the configuration of the neutral atom. For Na, it is 1s22s22p63s1.
Why is TI h2o 6 Coloured?
Ti(H2O)6 has unpaired electron for d-d transition so it is coloured . While Sc(H2O)6 can not undergo d-d Transition so it is colourles is colorless due to the absence of unpaired electrons.
Why is Sc and Zn colorless?
Thus Sc3+ and Ti4+ have completely empty d-orbitals and there are no electrons for the d-d transition thus they are colourless. Zn2+ has completely filled d-orbitals and there are no vacant d-orbitals for the transition of electrons , hence it is also colourless.