How many electrons are in energy levels?
.
Similarly one may ask, how many electrons are in the 4th energy level?
32 electrons
Beside above, how many electrons are in the 5th energy level? 2 electrons
Also to know, how are electrons found in the energy levels?
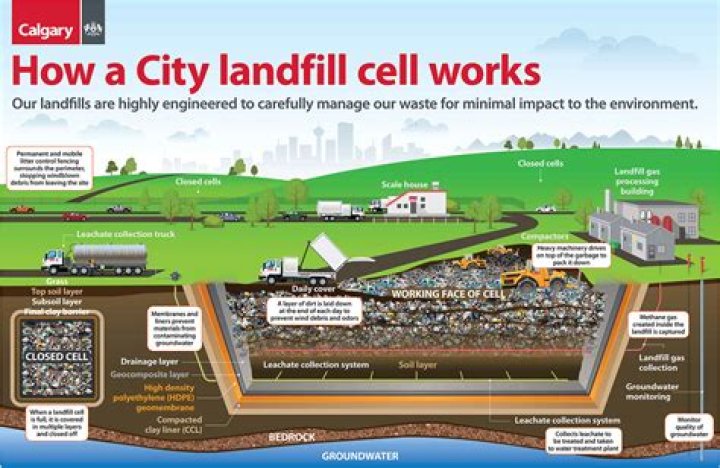
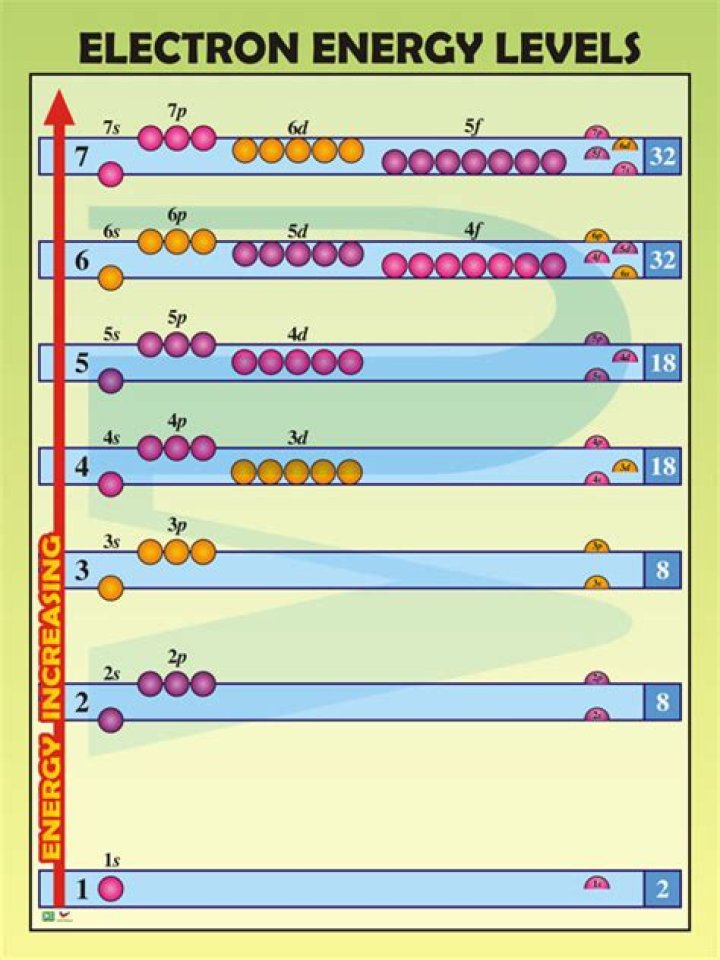
Summary. Energy levels (also called electron shells) are fixed distances from the nucleus of an atom where electrons may be found. As you go farther from the nucleus, electrons at higher energy levels have more energy. The maximum number of electrons at a given energy level depends on its number of orbitals.
Why can't electrons exist in between energy levels?
When an electron is hit by electricity, it can gain energy and can be bumped up to a higher energy orbit further away from the nucleus. On the other hand, when an electron loses energy, it falls back down to a lower energy orbit closer to the nucleus. The electron can never exist at distances in between allowed orbits.
Related Question AnswersHow are energy levels calculated?
Number of energy levels in each period- The atoms in the first period have electrons in 1 energy level.
- The atoms in the second period have electrons in 2 energy levels.
- The atoms in the third period have electrons in 3 energy levels.
- The atoms in the fourth period have electrons in 4 energy levels.
How many electrons does the 4th Shell hold?
The fourth energy level has 18 electrons. The fourth energy level of the periodic table includes the 4s 3d and 4p orbitals. The 4p orbital holds 6 electrons.How many electrons can the 7th Shell hold?
Notice that you can easily find the number of electrons that an atom can accomodate on a particular shell, or energy level, by using the fact that each orbital can hold a maximum of two electrons.How do you find the electrons?
The number of protons, neutrons, and electrons in an atom can be determined from a set of simple rules.- The number of protons in the nucleus of the atom is equal to the atomic number (Z).
- The number of electrons in a neutral atom is equal to the number of protons.
How many electrons can 2s hold?
The second shell has two subshells, s and p, which fill with electrons in that order. The 2s subshell holds a maximum of 2 electrons, and the 2p subshell holds a maximum of 6 electrons.What is the number of valence electrons in phosphorus?
fiveWhat is the highest energy level?
Electrons that are in the highest energy level are called valence electrons.Can electrons absorb any amount of energy?
An atom changes from a ground state to an excited state by taking on energy from its surroundings in a process called absorption. The electron absorbs the energy and jumps to a higher energy level. In the reverse process, emission, the electron returns to the ground state by releasing the extra energy it absorbed.What are electrons made of?
So in answer to the question "What are electrons made up of?" is, electrons are made up of charge, mass, and angular momentum. To make an electron, reach out across the electric field (which is otherwise just sitting there) and introduce enough wiggle to create −1.602×10−19 coulombs of charge.What is an energy level diagram?
Energy level diagrams are a means of analyzing the energies electrons can accept and release as they transition from one accepted orbital to another.Which electrons have the most energy?
Electrons at higher energy levels, which are farther from the nucleus, have more energy. They also have more orbitals and greater possible numbers of electrons. Electrons at the outermost energy level of an atom are called valence electrons.Are electrons positive or negative?
Proton—positive; electron—negative; neutron—no charge. The charge on the proton and electron are exactly the same size but opposite. The same number of protons and electrons exactly cancel one another in a neutral atom.How do you find the quantum number of electrons?
How to Determine the Number of Electrons With Quantum Numbers- Count the Full Orbitals.
- Add the Electrons for Each Full Orbital.
- Identify the Subshell Indicated by the Angular Quantum Number.
- Add the Electrons from the Full Subshells.
- Add the Electrons from Full Subshells to Those From Full Orbitals.
- Find the Legitimate Vales for the Magnetic Quantum Number.