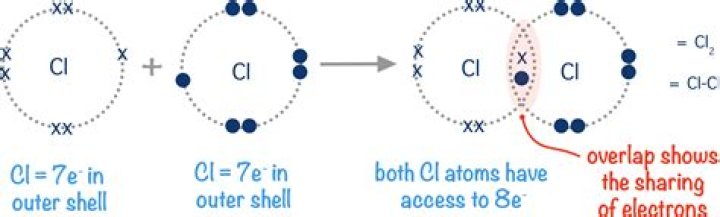
A covalent bond is pictured as two electrons shared by two different atomic nuclei, which means that each of the two nuclei has two electrons..
Beside this, how many electrons are present in a covalent bond?
2 electrons
One may also ask, how many electrons are in a triple covalent bond? A Triple bond is when three pairs of electrons are shared between two atoms in a molecule.
In this regard, how do you calculate covalent bonds?
The number of bonds for a neutral atom is equal to the number of electrons in the full valence shell (2 or 8 electrons) minus the number of valence electrons. This method works because each covalent bond that an atom forms adds another electron to an atoms valence shell without changing its charge.
What is covalent bond with example?
Covalent bonds are characterized by the sharing of electrons between two or more atoms. These bonds mostly occur between nonmetals or between two of the same (or similar) elements. Examples of compounds that contain only covalent bonds are methane (CH4), carbon monoxide (CO), and iodine monobromide (IBr).
Related Question Answers
Is Co a single covalent bond?
In these examples, each individual atom has just one unpaired electron to share with the other atom. When a covalent bond forms with one pair of shared electrons, we call it a single covalent bond. Methane, which is a carbon atom with single covalent bonds to four hydrogen atoms, is the simplest carbon compound.What is a polar covalent bond?
Polar covalent bonding is a type of chemical bond where a pair of electrons is unequally shared between two atoms. In a polar covalent bond, the electrons are not equally shared because one atom spends more time with the electrons than the other atom.What is meant by covalent bond?
A covalent bond, also called a molecular bond, is a chemical bond that involves the sharing of electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs, and the stable balance of attractive and repulsive forces between atoms, when they share electrons, is known as covalent bonding.Is water Polar?
A water molecule, because of its shape, is a polar molecule. That is, it has one side that is positively charged and one side that is negatively charged. The molecule is made up of two hydrogen atoms and one oxygen atom. The bonds between the atoms are called covalent bonds, because the atoms share electrons.What happens to electrons in an ionic bond?
Ionic bonding is the complete transfer of valence electron(s) between atoms. It is a type of chemical bond that generates two oppositely charged ions. In ionic bonds, the metal loses electrons to become a positively charged cation, whereas the nonmetal accepts those electrons to become a negatively charged anion.What is a polar bond?
Polar Bond Definition A polar bond is a covalent bond between two atoms where the electrons forming the bond are unequally distributed. This causes the molecule to have a slight electrical dipole moment where one end is slightly positive and the other is slightly negative.What are bonding electrons?
A bonding electron is an electron involved in chemical bonding. This can refer to: Covalent bond or molecular bond, a sharing of electron pairs between atoms. Bonding molecular orbital, an attraction between the atomic orbitals of atoms in a molecule.Is HBr a covalent bond?
HBr is a polar covalent bond.What type of bond is K and Cl?
Potassium atom has just one valence electron and Chlorine has 7 electrons, Cl atom needs just one more electron to complete its octet, which it receives from K atom. So an ionic bond forms between potassium and chlorine and Potassium Chloride gets formed.How many electrons are in a double covalent bond?
four electrons
How are hydrogen bonds formed?
A hydrogen bond is formed when the positive end of one molecule is attracted to the negative end of another. The concept is similar to magnetic attraction where opposite poles attract. Hydrogen has one proton and one electron. This makes hydrogen an electrically positive atom because it has a deficiency of electrons.What are the three types of covalent bonds?
The three types as mentioned in the other answers are polar covalent, nonpolar covalent, and coordinate covalent. The first, polar covalent, is formed between two nonmetals that have a difference in electronegativity. They share their electron density unevenly.Why are covalent bonds strong?
An atom that shares one or more of its electrons will complete its outer shell. Covalent bonds are strong - a lot of energy is needed to break them. Both nuclei are strongly attracted to the shared pair of electrons in the covalent bond, so covalent bonds are very strong and require a lot of energy to break.Is cl2 a single covalent bond?
A chlorine atom has 7 electrons in its valence shell—it needs 8 to complete it. Two chlorine atoms can share 1 electron each to form a single covalent bond. They become a Cl2 molecule. Oxygen can also form covalent bonds, however, it needs a further 2 electrons to complete its valence shell (it has 6).Which pair of elements will form a covalent bond?
Covalent bonds usually occur between nonmetals. For example, in water (H2O) each hydrogen (H) and oxygen (O) share a pair of electrons to make a molecule of two hydrogen atoms single bonded to a single oxygen atom. In general, ionic bonds occur between elements that are far apart on the periodic table.What is the difference between a sigma bond and a pi bond?
A sigma bond is your basic head-on covalent bond, with the bond in line with the bonding orbitals. You can only ever have one sigma bond between any two atoms. A pi bond is a covalent bond between orbitals perpendicular to the bond direction, usually p-orbitals (never s). A triple bond is one sigma and two pi bonds.Which is strongest bond?
Answer: Covalent bond is the strongest bond. Answer: There are a variety of ways atoms bond to one another.Can oxygen form a triple bond?
If oxygen starts with 6 electrons and forms a triple bond, then it has 2 lone pair electrons. Using the formal charge formula, 6 - (2+6/2) = 1. Since oxygen is so electronegative, it'll tend not to have a positive formal charge. However, oxygen can form triple bonds with carbon and nitrogen.Is HCl a covalent bond?
Answer and Explanation: HCl, also known as hydrochloric acid, has a covalent bond. The hydrogen (H) atom shares an electron with the chlorine (Cl) to form the bond.