How does density affect capillary action
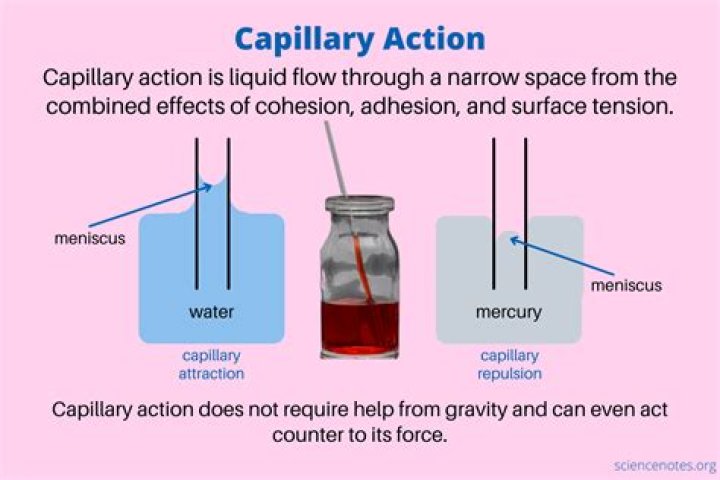
The more narrow the tube, the greater the rise of the liquid. … However, increased density of the liquid will cause it to rise to a lesser degree. The force with which water is held by capillary action varies with the quantity of water being held.
What factors influence capillary action?
Capillary action occurs because water is sticky, thanks to the forces of cohesion (water molecules like to stay close together) and adhesion (water molecules are attracted and stick to other substances).
How do you increase capillary action?
Increase capillary action: Increase temperature, decrease capillary tube diameter, perform any number of actions to decrease surface tension, etc…! Decrease capillary action: The opposite of the steps you would take to increase, also, increasing the density of the liquid you’re working with.
How does density affect surface tension?
After noting the density of each liquid it became clear that the higher the density the greater the surface tension. The experiment showed that water, the liquid with the highest density, has the highest surface tension; while alcohol,the liquid with the lowest density, has the lowest surface tension.Why does width affect capillary action?
Capillary action is demonstrated by the upward movement of water through a narrow tube against the force of gravity. … Capillary rise is the height to which the water rises within the tube, and decreases as the width of the tube increases. Thus, the narrower the tube, the water will rise to a greater height.
How does pressure affect capillary action?
The pressure at points just below the flat surface of the water in the surrounding vessel is atmospheric pressure. The system is, therefore not in equilibrium and the liquid rises in the capillary tube till the equilibrium is reached.
How does intermolecular forces affect capillary action?
Capillary Action. Intermolecular forces also cause a phenomenon called capillary action, which is the tendency of a polar liquid to rise against gravity into a small-diameter tube (a capillary), as shown in Figure 11.4. 3. When a glass capillary is is placed in liquid water, water rises up into the capillary.
Is surface tension is directly proportional to density?
The extent of the rise (or fall) is directly proportional to the surface tension of the liquid and inversely proportional to the density of the liquid and the radius of the tube.How surface tension is related to density and number of drops?
The ratio of any two drop volumes is almost equal to the ratio of the corresponding surface tension values.
Why is ice less dense than water?Ice actually has a very different structure than liquid water, in that the molecules align themselves in a regular lattice rather than more randomly as in the liquid form. It happens that the lattice arrangement allows water molecules to be more spread out than in a liquid, and, thus, ice is less dense than water.
Article first time published onHow does gravity affect capillary action?
Gravity causes the largest drops to flatten. Capillary rise with water: When a narrow tube is brought in contact with a mostly ‘wetting’ liquid, some of the liquid rises inside the tube. Capillary force supports the weight of the wetting film.
What are the factors affecting the capillary rise of water?
For this study, the factors used to determine capillary rise are the diameter of the capillary tube (representing the diameter of the pores in a soil), the contact angle between the liquid and the surface to which it adheres, the density of the liquid, the viscosity of the liquid, surface tension, and whether or not …
Why does diameter affect capillary rise?
It occurs because of intermolecular forces between the liquid and surrounding solid surfaces. If the diameter of the tube is sufficiently small, then the combination of surface tension (which is caused bycohesion within the liquid) and adhesive forces between the liquid and container act to lift the liquid.
How does surface tension affect capillary action?
The surface tension acts to hold the surface intact. Capillary action occurs when the adhesion to the surface material is stronger than the cohesive forces between the water molecules. The height to which capillary action will take water is limited by surface tension and gravity.
Why does the capillary rise decreases when the diameter is increased?
A thinner tube has less surface area and so the water molecules have less space to fill and that’s why they rise higher in a thin tube then a thick tube. This is the reason why the capilliary rise increase when the diameter of the tube decreases.
How does tube diameter affect capillary action?
If the diameter of the tube is sufficiently small, then the combination of surface tension (which is caused bycohesion within the liquid) and adhesive forces between the liquid and container act to lift the liquid.
How do different materials affect capillary action?
The thinner the tube, the higher the meniscus. Different types of materials (ie plastic or glass) can affect the result. Relating it to infill… material such as small, angular sand with small void spaces will have stronger capillary action.
What is the relation between capillary rise and diameter of the capillary tube?
Capillary rise is inversely proportional to the capillary diameter.
Is capillary pressure high or low?
Capillaroscopy has been used to visualize capillaries in the skin in 2D, and has been reported to observe an average range of capillary pressure of 10.5 to 22.5 mmHg in humans, and an increase in pressure among people with type 1 diabetes and hypertension.
Does angle affect capillary action?
From this, we see that the lower the contact angle (the more hydrophilic the pore) the higher the capillary pressure and thus the the stronger the capillary action.
Is the capillary rise greater in small or large diameter tubes?
The smaller the diameter of the tube, the higher it will rise.
What are factors affecting surface tension?
Surface tension is caused by the effects of intermolecular forces at the interface. Surface tension depends on the nature of the liquid, the surrounding environment and temperature. Liquids where molecules have large attractive intermolecular force will have a large surface tension.
What is the effect of surface tension on number of drops?
The experimental results show that a lower surface tension will result in a higher tangential spread factor and a lower receding rate during the receding stage, for the drop impinging and depositing on a rotating disk. In addition, a lower surface tension of the drop tends to promote the occurrence of splash.
What is the relationship between surface tension and drop diameter?
It is concluded that surface tension can be expected to decrease with decrease in droplet size over a wide range of circumstances. In addition, approximate figures are obtained for the rate at which such decreases may be expected. The decreases become significant for very small drops.
Why surface tension decreases with increase in concentration?
The surface tension decreases with increase in concentration. This is due to breaking of hydrogen bonds when surfactants are added in water, leading to higher adsorption at the air-water interface. … With increase in temperature the surface tension decreased steadily.
What is surface energy density?
The surface energy density is: (f dx) / (L dx) = f/L = γ , i.e. equal to the surface tension. … A sphere is the geometric form which has the smallest surface area for a given volume. Thus it is clear that the surface of the liquid must have a higher energy than the bulk.
Why interfacial tension is less than surface tension?
Interfacial tension (IFT) arises from differences between the intermolecular forces at the two liquid surfaces in contact with each other. … Therefore, the surface has excess Gibbs energy relative to the interior of the liquid. Surface tension is the excess energy per unit area (force per unit length; SI unit is Nm-1).
How does density compared to water?
Key Concepts. Density is a measure of how heavy something is compared to its size. If an object is more dense than water it will sink when placed in water, and if it is less dense than water it will float. Density is a characteristic property of a substance and doesn’t depend on the amount of substance.
What is meant by density?
density, mass of a unit volume of a material substance. … Density offers a convenient means of obtaining the mass of a body from its volume or vice versa; the mass is equal to the volume multiplied by the density (M = Vd), while the volume is equal to the mass divided by the density (V = M/d).
Why is the density of water important?
Density has obvious importance when it comes to the buoyancy of objects. Broadly, if something is denser than water (having a density over 1,000 kg/cubic meter) it will sink, but if something has a lower density than water, it will float. … The difference in density is also why oil floats on the surface of water.
Why does water rise up a capillary tube?
Capillarity is the result of surface, or interfacial, forces. The rise of water in a thin tube inserted in water is caused by forces of attraction between the molecules of water and the glass walls and among the molecules of water themselves. … The narrower the bore of the capillary tube, the higher the water rises.