How do you know if something is an aldehyde
Aldehydes contain the carbonyl group bonded to at least one hydrogen atom. … The carbon atom of this group has two remaining bonds that may be occupied by hydrogen, alkyl or aryl substituents. If at least one of these substituents is hydrogen, the compound is an aldehyde.
How do you identify an aldehyde in a lab?
Take the given organic compound to be tested in a clean test tube. Add 2-3 drops of Schiff’s reagent. If there is instant pink or red colour formation then the presence of aldehyde is confirmed.
What is the general test for the presence of aldehydes?
Tollen’s reagent is a classical organic laboratory technique to test for the presence of an aldehyde. The reagent consists of silver(I) ions dissolved in dilute ammonia. When the aldehyde is oxidized, the silver(I) ions are reduced to silver metal.
How do you distinguish between aldehydes and ketones?
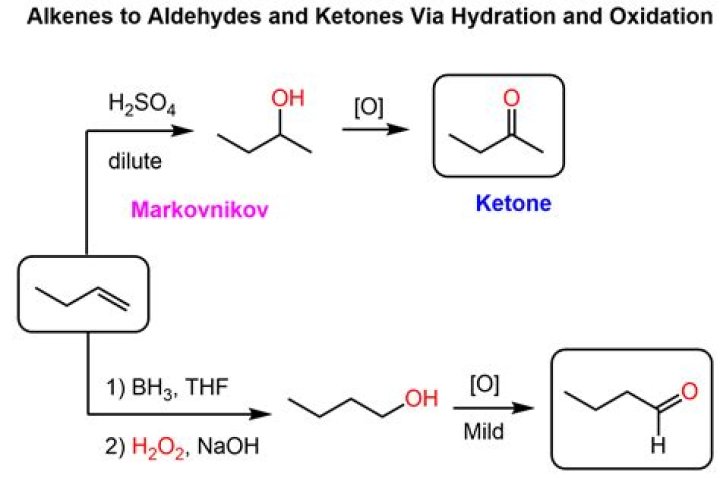
You will remember that the difference between an aldehyde and a ketone is the presence of a hydrogen atom attached to the carbon-oxygen double bond in the aldehyde. Ketones don’t have that hydrogen. The presence of that hydrogen atom makes aldehydes very easy to oxidize (i.e., they are strong reducing agents).What is an example of an aldehyde?
Examples are retinal (vitamin A aldehyde), important in human vision, and pyridoxal phosphate, one of the forms of vitamin B6. Glucose and other so-called reducing sugars are aldehydes, as are several natural and synthetic hormones.
How can you tell the difference between an alcohol and an aldehyde?
Precipitates that are yellow indicate non-conjugated ketones or aldehydes, whereas red-orange precipitates indicate conjugated systems. This test is used to differentiate ketones and aldehydes from alcohols and esters with which DNPH does not react, and thus no precipitate is formed.
How do you identify an aldehyde functional group?
The location of the carbonyl group in the molecule is identified by numbering the longest chain of carbons so that the carbonyl group has the lowest number possible. When one of the groups attached to the carbonyl group is a hydrogen instead of a carbon, the compound is called an aldehyde.
Is glucose an aldehyde?
Glucose and fructose, for example, are carbohydrates with the formula C6H12O6. These sugars differ in the location of the C=O. double bond on the six-carbon chain, as shown in the figure below. Glucose is an aldehyde; fructose is a ketone.Which test is not given by acetaldehyde?
Acetaldehyde cannot show Lucas test because Lucas test is given by alcohols only. It is used in the distinction between primary, secondary and tertiary alcohols.
Which test is used to identify the aldehyde and ketone?Tollens’ test, also known as silver-mirror test, is a qualitative laboratory test used to distinguish between an aldehyde and a ketone. It exploits the fact that aldehydes are readily oxidized (see oxidation), whereas ketones are not.
Article first time published onWhat are the characteristics of a positive tollens test for aldehydes?
In a positive test, the diamine silver(I) complex oxidizes the aldehyde to a carboxylate ion and in the process is reduced to elemental silver and aqueous ammonia. The elemental silver precipitates out of solution, occasionally onto the inner surface of the reaction vessel, giving a characteristic “silver mirror”.
What reagent is used to test for aldehydes?
Tollens’ reagent is an alkaline solution of ammoniacal silver nitrate and is used to test for aldehydes. Silver ions in the presence of hydroxide ions come out of solution as a brown precipitate of silver(I) oxide, Ag2O(s).
Is acetone an aldehyde?
Propanone (acetone)Butanone (methyl ethyl ketone)AcetophenoneBenzophenone
What are aldehydes in food?
Aldehydes are characteristic compounds of secondary oxida- tion in the autoxidative process of fats, oils, lipidic foods and biolo- gical membranes. … Cis-3- hexenal and 2,4-pentadienal are involved in the soybean oil flavor. Hexanal and 2-hexenal cause the “unripe odors” in some fruits and vegetables.
What is the example of aldehyde used in making Bakelite?
The plastic Bakelite is made by a reaction between formaldehyde and phenol. It is not a linear chain but has a three dimensional structure. Similar three-dimensional polymers are made from formaldehyde and the compounds urea and melamine.
What are the properties of aldehyde?
Aldehydes have properties that are diverse and that depend on the remainder of the molecule. Smaller aldehydes are more soluble in water, formaldehyde and acetaldehyde completely so. The volatile aldehydes have pungent odors.
Do aromatic aldehydes give Schiff's test?
Schiff’s reagent A reagent used for testing for aldehydes and ketones; it consists of a solution of fuchsin dye that has been decolorized by sulphur dioxide. … Aromatic aldehydes and aliphatic ketones restore the colour slowly. It is named after the German chemist Hugo Schiff (1834–1915).
What is Brady's reagent used for?
Brady’s reagent or Borche’s reagent, is prepared by dissolving 2,4-dinitrophenylhydrazine in a solution containing methanol and some concentrated sulfuric acid. This solution is used to detect ketones and aldehydes.
How can you tell the difference between acetone and acetaldehyde?
Acetone is the smallest member of the ketone group, whereas acetaldehyde is the smallest member of aldehyde group. The key difference between Acetaldehyde and Acetone is the number of carbon atoms in the structure; acetone has three Carbon atoms, but acetaldehyde has only two carbon atoms.
Which reagent is used to distinguish between formaldehyde and acetaldehyde?
Complete answer: On treatment of formaldehyde and acetaldehyde with iodine in the presence of base, acetaldehyde gives off yellow color precipitation while formaldehyde does not react with it. This is known as iodoform reaction and the test is called iodoform test.
Which of the aldehyde is most reactive?
Thus, acetaldehyde is the most reactive compound which is option ‘d’.
Is propanol an aldehyde?
Propionaldehyde or propanal is the organic compound with the formula CH3CH2CHO. It is the 3-carbon aldehyde. It is a colourless, flammable liquid with a slightly fruity odour.
Is sucrose an aldehyde?
Sucrose (glucose + fructose) lacks a free aldehyde or ketone group and therefore is non-reducing.
Does dextrose have an aldehyde?
Glucose is the most widely distributed carbohydrate. It is also called dextrose because it is dextrorotatory, meaning that it rotates the plane of plane-polarized light in the (+) direction. It has molecular formula C6H12O6 and is structurally a pentahydroxy aldehyde (Fig. 10-1).
Do aromatic aldehydes give tollens test?
Tollens test is given by aldehydes including both aromatic and aliphatic aldehydes. Whereas fehlings is reduced by only aliphatic aldehyde. Aromatic aldehydes do not show this reaction.
Can Fehling test be used to distinguish between an aldehyde and a ketone?
Fehling’s solution can be used to distinguish aldehyde vs ketone functional groups. The compound to be tested is added to the Fehling’s solution and the mixture is heated. Aldehydes are oxidized, giving a positive result, but ketones do not react, unless they are α-hydroxy ketones.
What is tollens reagent explain its reaction with aldehydes?
Tollens Reagent : Freshly prepared ammonical silver nitrate solution is called Tollens reagent. → On warming an aldehyde with Tollens reagent a bright silver mirror is produce due to formation of silver metal. R-CHO+2[Ag(NH3)2]++3OH-→RCOO-+2Ag+2H2O+4NH3.
Is acetone a methyl ketone?
A methyl ketone that consists of propane bearing an oxo group at C2. 1 model in this collection. It is the simplest and smallest ketone. …
Is Brady's test also positive for ketones like aldehydes?
How to Prepare Brady’s Reagent. Brady’s reagent is a red to orange solid of DNPH, methanol, and sulfuric acid that reacts with carbonyl compounds such as aldehydes and ketones. … A positive test is signaled by a yellow precipitate, for aliphatic carbonyls, or red to orange precipitate, for aromatic carbonyls.
Is tollens reagent explosive?
Tollen’s reagent should never be prepared in advance as ammoniacal silver solutions are hazardous and can explode on standing. Any unused solution should be deactivated as soon as possible and within two hours.
Is ethanol an aldehyde?
You get an aldehyde if you use an excess of the alcohol, and distil off the aldehyde as soon as it forms. … If you used ethanol as a typical primary alcohol, you would produce the aldehyde ethanal, CH3CHO.