Plasma calcium levels in mammals are tightly regulated, with bone acting as the major mineral storage site. Calcium ions, Ca2+, are released from bone into the bloodstream under controlled conditions. Calcium is transported through the bloodstream as dissolved ions or bound to proteins such as serum albumin..
People also ask, what is the formula for the calcium ion?
Calcium ion | Ca+2 - PubChem.
Beside above, how many ions does calcium have? Calcium, Ca It has two electrons in its outer shell. When these electrons are lost, a calcium ion, Ca 2+, is formed. A calcium ion has the same electronic structure as an argon atom (Ar).
Considering this, what is the most common ion of calcium?
The calcium ion is also known as Ca2+, meaning it has two less electrons than protons and is not stable in nature.
Where calcium is found?
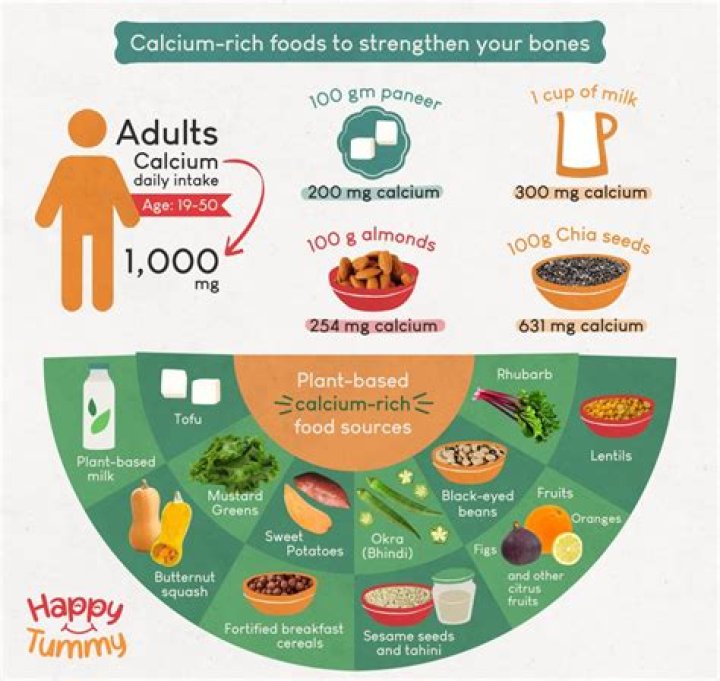
In nature, calcium is found in rocks, chalk, and seashells. It is found in the bones, muscles, and nerves of animals and in many different plants. In the food you eat, there is a lot of calcium in milk and other dairy products. There is also a good deal of calcium in green leafy vegetables like spinach.
Related Question Answers
What are calcium ions needed for?
Calcium ions (Ca2+) contribute to the physiology and biochemistry of organisms cell. They play an important role in signal transduction pathways, where they act as a second messenger, in neurotransmitter release from neurons, in contraction of all muscle cell types, and in fertilization.What is the difference between a calcium atom and a calcium ion?
For example, a neutral calcium atom, with 20 protons and 20 electrons, readily loses two electrons. This results in a cation with 20 protons, 18 electrons, and a 2+ charge. The name of a metal ion is the same as the name of the metal atom from which it forms, so Ca2+ is called a calcium ion.What do calcium ions do in muscle contraction?
Muscle contraction: Calcium remains in the sarcoplasmic reticulum until released by a stimulus. Calcium then binds to troponin, causing the troponin to change shape and remove the tropomyosin from the binding sites. Cross-bridge cling continues until the calcium ions and ATP are no longer available.What does +2 charge mean?
Ions. When an atom gains or loses electrons, it becomes a charged particle known as an ion. When an atom gains extra electrons, it becomes a negatively charged ion. If it has only one extra electron, its charge is -1. If it gains two extra electrons, its charge is -2, and so on.What does Ca+ mean?
Medical Definition of CA CA: 1. Short (and slang) for cancer and carcinoma. 2. Abbreviation for cardiac arrest; chronological age; coronary artery.What helps calcium absorption?
In addition to vitamin D, vitamin C, vitamin E, vitamin K, magnesium, and boron assist in absorbing calcium and also increasing bone mass. Exercise also helps the body absorb calcium. Because the body has a hard time absorbing a large amount of calcium at once, spreading out the intake of calcium is recommended.What is the name of mg2+?
Magnesium ion | Mg+2 - PubChem.Is calcium a cation?
A cation is an atom or molecule in which the protons outnumber the electrons and hence create a positive charge. Common cations include sodium, potassium, calcium, magnesium, copper, iron, and mercury. The cations of greatest importance in anaesthesia and intensive care are sodium, potassium, calcium, and magnesium.What are the most common ions in the human body?
Answer and Explanation: The fours most abundant ions in the body are potassium, sodium, calcium, and chloride.What is the most common ion of aluminum?
Aluminum is in the fifth column and therefore has 5 electrons in its outermost shell. It would tend to lose three electrons and form a +3 ion. Magnesium is in the second column and therefore has 2 electrons in its outermost shell.Is calcium an anion or cation?
Nomenclature of simple ions Rule 1: Cations. Naming the element and adding the word “ion” forms the cation name. So, Na+ is “sodium ion”. Ca+2 is “calcium ion”.What is the most common ion of fluorine?
Fluorine is in Group 7. It has seven electrons in its outer shell. It gains an electron from another atom in reactions, forming a fluoride ion, F -. Note that the atom is called fluorine, but the ion is called fluoride.What is the most common ion of chlorine?
chloride ion
How many protons does a calcium ion have?
20
Why do ions form?
Ions are formed when atoms lose or gain electrons in order to fulfill the octet rule and have full outer valence electron shells. When they lose electrons, they become positively charged and are named cations. When they gain electrons, they are negatively charged and are named anions.What is an ion in chemistry?
An ion is a charged atom or molecule. When an atom is attracted to another atom because it has an unequal number of electrons and protons, the atom is called an ION. If the atom has more electrons than protons, it is a negative ion, or ANION. If it has more protons than electrons,it is a positive ion.What is meant by covalent bond?
A covalent bond, also called a molecular bond, is a chemical bond that involves the sharing of electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs, and the stable balance of attractive and repulsive forces between atoms, when they share electrons, is known as covalent bonding.How are ionic compounds formed?
SUMMARY PRINCIPLE OF IONIC COMPOUNDS An ionic compound is formed by the complete transfer of electrons from a metal to a nonmetal and the resulting ions have achieved an octet. The protons do not change. Metal atoms in Groups 1-3 lose electrons to non-metal atoms with 5-7 electrons missing in the outer level.How does an ionic bond form?
Ionic bond, also called electrovalent bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Such a bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom.