How do you convert alkane to haloalkane?
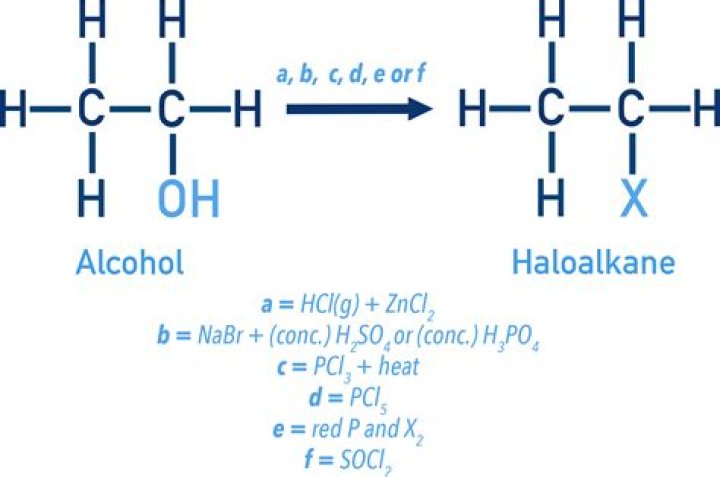
How do you convert alkane to haloalkane?
The reaction of a halogen with an alkane in the presence of ultraviolet (UV) light or heat leads to the formation of a haloalkane (alkyl halide). An example is the chlorination of methane.
Can Haloalkanes undergo free radical substitution?
Explanation: Alkanes can be converted to Halogenalkanes through a free radical substitution as free radicals are highly reactive. Lets use the reaction between Chlorine and Methane ( CH4 ), which can occur in the atmosphere. Cl2→2Cl.
What is free radical substitution reaction in alkanes?
In organic chemistry, free-radical halogenation is a type of halogenation. This chemical reaction is typical of alkanes and alkyl-substituted aromatics under application of UV light. The reaction is used for the industrial synthesis of chloroform (CHCl3), dichloromethane (CH2Cl2), and hexachlorobutadiene.
What type of reaction is alkane to haloalkane?
Alkanes (the most basic of all organic compounds) undergo very few reactions. The two reactions of more importaces is combustion and halogenation, (i.e., substitution of a single hydrogen on the alkane for a single halogen) to form a haloalkane.
What is Haloalkane and Haloarene?
Haloalkanes contain halogen atom(s) attached to the sp3 hybridised carbon atom of an alkyl group whereas haloarenes contain halogen atom(s) attached to sp2 hybridised carbon atom(s) of an aryl group. Many halogen containing organic compounds occur in nature and some of these are clinically useful.
What is secondary Haloalkane?
A secondary halogenoalkane has the halogen bonded to a carbon that is itself attached to two other carbon atoms. In tertiary halogenoalkanes, the halogen is bonded to a carbon that is itself attached to three other carbon atoms. This is a useful reaction for increasing the chain length by one carbon atom.
What happens when alkane undergoes free radical substitution and halogenation?
Alkanes (the most basic of all organic compounds) undergo very few reactions. One of these reactions is halogenation, or the substitution of a single hydrogen on the alkane for a single halogen to form a haloalkane.
Which step in the radical chlorination of an alkane is the rate controlling step?
bromination
So, in the case of bromination, the formation of the radical is the rate-determining step, the slowest step in our reaction. And it has the highest activation energy.
Which of the following can bring about free radical chlorination of propane?`?
Breaking A Secondary C–H Bond Results In A More Stable Free Radical. Let’s turn to propane. In the free radical chlorination reaction, chlorine radical may abstract a hydrogen from either from one of the methyl groups of propane, or from the methylene.
What is difference between alkane and haloalkane?
is that alkane is (organic chemistry) any of saturated hydrocarbons including methane, ethane and compounds with long carbon chain known as paraffins etc, having a chemical formula of the form cnh2n+2 while haloalkane is (organic chemistry) any alkane in which one or more hydrogen atoms has been replaced by that of a …
How will you differentiate between haloalkane and Haloarene?
Haloalkanes are hydrocarbons containing aliphatic alkane with one or more hydrogen atom/s replaced by halogens. Haloarenes are hydrocarbons containing aromatic alkane with one or more hydrogen atom/s replaced by halogens. 2. Haloalkanes are aliphatic hydrocarbons.
Which condensed formula represents a Haloalkane?
The general formula for a primary haloalkane (1° alkyl halide) is R-CH2X where R is an alkyl chain and X is the halogen atom.