When you put a piece of aluminium in acid to remove the oxide layer. Take it out and put in acetone and then ether. You will for a very short time see pure Aluminium. It is highly electropositive and will react with oxygen very quickly to form Aluminium oxide..
Also, when aluminum and oxygen combine the formula is?
This means that the chemical formula for aluminum oxide is simply Al2 O3. That's 2 aluminum atoms for every 3 oxygen atoms.
Additionally, how does Aluminium react with air? Reaction of aluminium with air Aluminium reacts with oxygen, forming a protective layer of alumnium(III) oxide that prevents further reaction with oxygen. Like magnesium, aluminium burns in oxygen with a brilliant white flame. The product in this reaction is also alumnium(III) oxide.
In respect to this, what happens when Aluminium reacts with chlorine?
When aluminium metal comes in contact with chlorine gas in presence of heat, violent reaction get started and Aluminium chloride powder/granule forms. This is an exothermic reaction. After starting the reaction and due to exothermic reaction elevated temperature helps in auto reaction in-between aluminium and Chlorine.
What happens when you mix oxygen and potassium?
Potassium oxide is produced from the reaction of oxygen and potassium; this reaction affords potassium peroxide, K2O2. Potassium hydroxide cannot be further dehydrated to the oxide but it can react with molten potassium to produce it, releasing hydrogen as a byproduct.
Related Question Answers
What is the reaction between aluminum and oxygen?
Reaction of aluminium with air So, normally, aulumium metal does not react with air. If the oxide layer is damaged, the aluminium metal is exposed to attack. Aluminium will burn in oxygen with a brilliant white flame to form the trioxide alumnium(III) oxide, Al2O3.What is the charge of aluminum?
The charge of an aluminum ion is typically 3+. This is because the element's atomic number is 13, reflecting the fact that it has 13 electrons and 13 protons. The valence shell of aluminum has three electrons, and per the octet rule, these three electrons are lost resulting in just 10 electrons and 13 protons.Is aluminum and oxygen a covalent bond?
we know that aluminium has +3 charge while oxygen has -2, according to rule if the oxidation state of element isn`t same they are interchanged so by following this rule we can say that aluminium oxide has ionic character (Al2O3). this also shows covalent nature because of its small Atomic Radi.How do you combine two reactions?
You combine multiple reactions into a single equation by listing all the reactants on the left side of the equation and all the products on the right side of the equation. Simplification of the overall equation will eliminate chemical species that exist on both sides of the equation without change.How do you solve a double replacement reaction?
How to Complete a Double Displacement Reaction - Step 1: Identify the Individual Ions from the Reactants and Their Charges. For the reactant Na 2 S , there is a cation (positive ion) and an anion (negative ion).
- Step 2: Switch the Cations and Anions of the reactants.
- Step 3: Balance the Chemical Reaction.
What is the correct name for Al2O3?
Aluminum oxide
Is Rusting a combination reaction?
Rust is the product of a combination reaction of iron and oxygen: Since this new compound is formed from a metal and a non-metal, it is an ionic compound.Does aluminum react with chlorine?
"Aluminum and chlorine do not react rapidly at room temperature, but if the aluminum is heated, the reaction is vigorous. The product is aluminum chloride, a white powder that fumes in air, reacting with water to form HCl gas and aluminum oxide."What happens when fluorine reacts with Aluminium?
Aluminum react with fluorine to produce aluminum fluoride. This reaction takes place at a temperature near 600°C.What does aluminum react with?
Aluminum Reactions Aluminium reacts with dilute hydrochloric acid to form aluminium chloride and hydrogen gas. Chlorine and liquid bromine react with aluminum at room temperature.What state of matter is aluminum chloride?
Aluminium chloride (AlCl3), also known as aluminium trichloride, is the main compound of aluminium and chlorine. It is white, but samples are often contaminated with iron(III) chloride, giving it a yellow color. The solid has a low melting and boiling point.What type of bond is aluminum chloride?
Explanation: The Al-Cl bond is polar covalent. This is consistent with the fact that aluminum chloride changes directly from a solid to a gas at the relatively low temperature of 180 °C. Al has three valence electrons.Why Aluminium does not react with dilute Sulphuric acid?
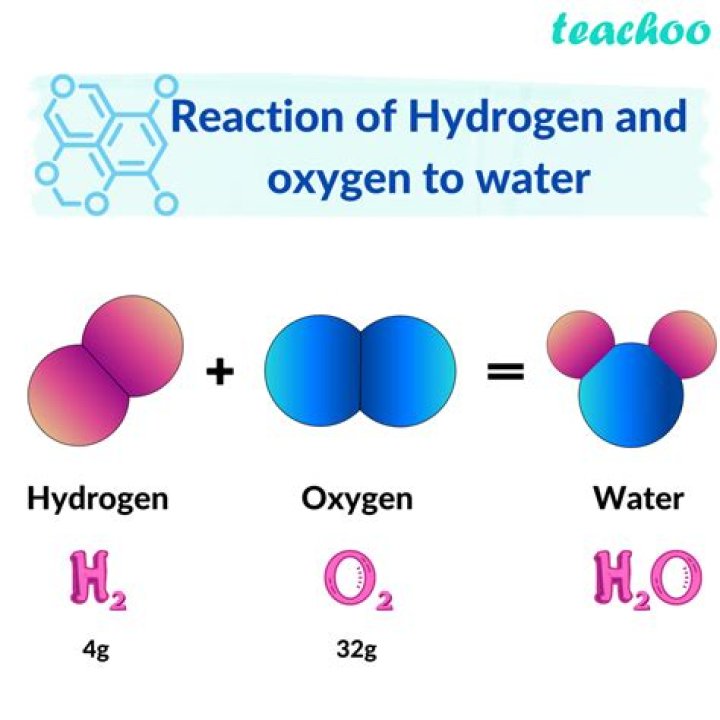
If PURE Aluminium is Placed in Cold Dilute H2SO4 it will Slowly Dissolve into a Whitish Solution which if Dried will Form Aluminium Sulphate Crystals as a Fine Powder !!!!! The Reaction will Evolve Hydrogen Gas which will Combine with Oxygen to form Water ! The Reaction is Not Very Exciting and Nothing Much Happens !!Does aluminum dissolve in sulfuric acid?
Aluminium metal dissolves easily in dilute sulfuric acid to form an aqueous solution with the aluminium ion III, Al3 +, the sulfate ion, SO42-, and hydrogen gas, H2.Why does Aluminium not react with steam?
Aluminium is unusual, because it is a reactive metal that does not react with water. Its surface forms a protective layer of aluminium oxide that keeps water away from the metal below. The apparatus used to react a metal with steam and collect the gas produced is shown below.Is aluminum toxic to humans?
Oral exposure to aluminum is usually not harmful. Some studies show that people exposed to high levels of aluminum may develop Alzheimer's disease, but other studies have not found this to be true. The kidney disease causes less aluminum to be removed from the body in the urine.What happens when aluminum reacts with hydrochloric acid?
Explanation: Aluminium metal will react with dilute hydrochloric acid to produce aqueous aluminium chloride, AlCl3 , and hydrogen gas, H2 . Keep in mind that this reaction will not take place as soon as you add the piece of aluminium to the hydrochloric acid solution.Why is aluminum resistant to air and water?
Aluminum oxide consists of atoms of aluminum and oxygen bonded together. structure changes just enough to become chemically inert and thus unable to react rapidly with additional water molecules or atmospheric oxygen. This change in molecular structure is why aluminum oxide metal resists corrosion.