How are Skittles used in chromatography?
How are Skittles used in chromatography?
Start by choosing one of each color of Skittles, and placing them in a line equally spaced apart on the foil. Dip your finger into some water and make a small drop next to each candy. Place one Skittle in each drop, and let sit for about a minute to let the color come off. Afterwards, you can eat the candy.
What is the purpose of the candy chromatography lab?
Colorful candies like Skittles are made with FD&C dyes, sugar and other substances. Chromatography is a technique used to separate different parts of a solution. It is used to identify chemicals, used to identify crime scene samples like blood or drugs and used to demonstrate the different dyes in your favorite candy.
Which dye travels furthest up a chromatogram?
polar dye
The most polar dye is the dye will move farthest up the paper when water is the solvent; the least polar dye will move farthest up the paper when alcohol is the solvent.
How does candy get its color?
Some candy colors are the result of a single food coloring whereas others may use a combination of dyes to create just the right appearance. In this activity you’ll get to do some scientific detective work at home to investigate what different dyes are used to color M&M candies.
Are the pigments in green M&Ms and green Skittles the same?
The color of the regular M&M is more of a true green, while the color of a green Skittle is a more yellow green. If you opted to taste the green colors, you probably found that the Skittles dye tasted more sour, probably because the sour Skittle’s interior was also dissolving.
Does the color of the candies mix?
Most Candy = Artificial Colors Just like when you’re mixing paints in art, it’s takes a combination of dyes to create certain colors. Colors get mixed up, but did you know you can separate them again?
Do you think the pigments in green M&M’s and green Skittles are the same?
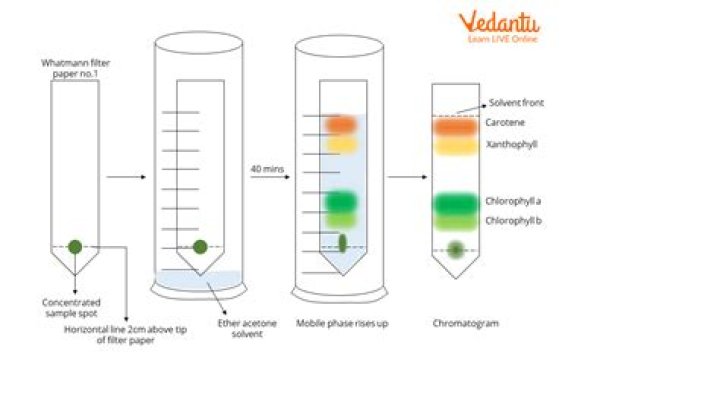
How do Colours separate in chromatography?
Often the colors that we see are a combination of the light reflected by a mixture of different-color molecules. Different molecules run up the paper at different rates. As a result, components of the solution separate and, in this case, become visible as strips of color on the chromatography paper.
Which dye color is most polar?
Red 40 dye is somewhat more polar than Blue 1 dye. Each molecule will have a similar amount of charge from ionization, but since Blue 1 is a larger molecule, the resulting charge will be spread out over a larger molecule.
Which M&M color dissolves the faster in water?
Did you notice that different colors dissolved at different rates? The red M&M dissolved the fastest and the blue M&M dissolved the slowest.



