Acyl chlorides generally react rapidly (even violently) with hydroxide ions from, say, sodium hydroxide solution. Again, if the -COCl group is attached to a benzene ring, the reactions are slower..
Consequently, what do acyl chlorides react with?
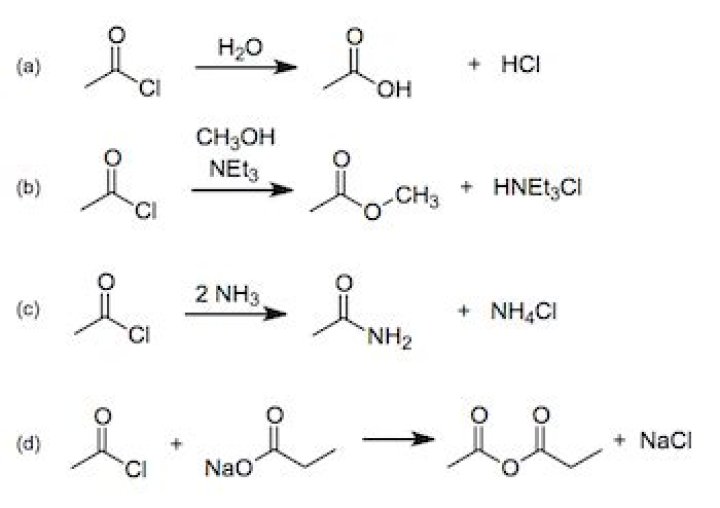
Acid chlorides react with carboxylic acids to form anhydrides. Acid chlorides react with water to form carboxylic acids. Acid chlorides react with ammonia, 1o amines and 2o amines to form amides.
Also, do acyl chlorides react with silver nitrate? Reactions of Acyl Chlorides. Acyl Chlorides react at room temperature with ammonia to form amides. This reaction is very vigorous and the products form white precipitate of silver chloride when silver nitrate is added, immediately.
In respect to this, can tertiary amine react with acyl chloride?
Tertiary amines CANNOT produce amides by reaction with acyl chlorides but their high basicity allow them to capture a molecule of HCl and transform acyl chlorides into KETENES (R-C=C=O). IMPORTANT: For this reaction to proceed it is imprescindible that the acyl chloride have at least one alpha hydrogen.
What happens when ethanol reacts with acetyl chloride?
Ethanoyl chloride reacts instantly with cold ethanol. There is a very exothermic reaction in which a steamy acidic gas is given off (hydrogen chloride). Ethyl ethanoate (an ester) is formed.
Related Question Answers
How do you convert carboxylic acid to acyl chloride?
Carboxylic acids react with Thionyl Chloride (SOCl2) to form acid chlorides. During the reaction the hydroxyl group of the carboxylic acid is converted to a chlorosulfite intermediate making it a better leaving group. The chloride anion produced during the reaction acts a nucleophile.Why do acyl chlorides react faster?
Acyl chlorides are the most reactive carboxylic acid derivatives. The electronegative chlorine atom pulls electrons toward it in the C-Cl bond, which makes the carbonyl carbon more electrophilic. This makes nucleophilic attack easier. Also, the Cl- is an excellent leaving group, so that step is also fast.How do you name an acid chloride?
Naming acyl halides - The easiest way to name an acyl halide is to consider the relationship with the corresponding carboxylic acid:
- If something is substituted into the hydrocarbon chain, the carbon in the -COX (X = halide) group is the number 1 carbon.
- For example, 2-methylbutanoyl chloride is named as follows:
Why acyl chloride is more reactive than carboxylic acids?
Acyl chlorides are more reactive than carboxylic acids or carboxylic esters because they have a good leaving group attached to the carbonyl carbon.How do you make an acid chloride?
Acid chlorides can be formed from the reaction between a carboxylic acid and thionyl chloride. Additionally, acid chlorides can be used in the reaction with a carboxylic acid to make anhydride compounds.How do you identify acyl chloride?
An acyl chloride like ethanoyl chloride is a colourless fuming liquid. The strong smell of ethanoyl chloride is a mixture of the smell of vinegar (ethanoic acid) and the acrid smell of hydrogen chloride gas. The smell and the fumes are because ethanoyl chloride reacts with water vapour in the air.Why is formyl chloride unstable?
1 Answer. The instability of HCOCl is caused by ease of elimination of HCl from its molecules. Cl is decent leaving group and after it's gone, remaining acylium cation has a very acidic hydrogen instead of alkyl present in other acyl halides.What does acylation mean?
In chemistry, acylation (or alkanoylation) is the process of adding an acyl group to a compound. The compound providing the acyl group is called the acylating agent. Acylation can be used to prevent rearrangement reactions that would normally occur in alkylation.What type of compound is formed when an amine reacts with an acyl chloride?
The reaction with ammonia So in the first instance you get hydrogen chloride gas and an organic compound called an amide. Amides contain the group -CONH2. In the reaction between ethanoyl chloride and ammonia, the amide formed is called ethanamide.Which amine does not react with acetyl chloride?
Tertiary amine do not react with acetyl chloride.Why is Ethanoic anhydride used instead of Ethanoyl chloride?
The reasons for using ethanoic anhydride rather than ethanoyl chloride include: Ethanoic anhydride is safer to use than ethanoyl chloride. It is less corrosive and not so readily hydrolysed (its reaction with water is slower).Why do amines act as bases?
15.13 Amines as Bases Recall that ammonia (NH 3) acts as a base because the nitrogen atom has a lone pair of electrons that can accept a proton. Nearly all amines, including those that are not very soluble in water, will react with strong acids to form salts soluble in water.Are acid chlorides acidic?
3 Answers. An acid chloride is a covalent molecule. It has no pH. An acid chloride is an oxo-acid whose acidic −OH has been replaced with −Cl.Why is Ethanoyl chloride a good acylating agent?
Ethanoyl chloride is more reactive than ethanoic anhydride and fumes in moist air. The fumes are due to hydrogen chloride gas condensing water droplets forming a mist of hydrochloric acid.What is an anhydride bond?
An acid anhydride is a compound that has two acyl groups bonded to the same oxygen atom. A common type of organic acid anhydride is a carboxylic anhydride, where the parent acid is a carboxylic acid, the formula of the anhydride being (RC(O))2O.Is acid chloride esterification reversible?
Dry hydrogen chloride gas is used in some cases, but these tend to involve aromatic esters (ones containing a benzene ring). If you are a UK A level student you won't have to worry about these. The esterification reaction is both slow and reversible.Is esterification addition elimination?
In chemistry, an addition-elimination reaction is a two-stage reaction process of an addition reaction followed by an elimination reaction. This gives an overall effect of substitution, and is the mechanism of the common nucleophilic acyl substitution often seen with esters, amides, and related structures.What is the product when Ethanoyl chloride is reacted with propanol?
Ethanoyl chloride reacts instantly with cold ethanol. There is a very exothermic reaction in which a steamy acidic gas is given off (hydrogen chloride). Ethyl ethanoate (an ester) is formed.