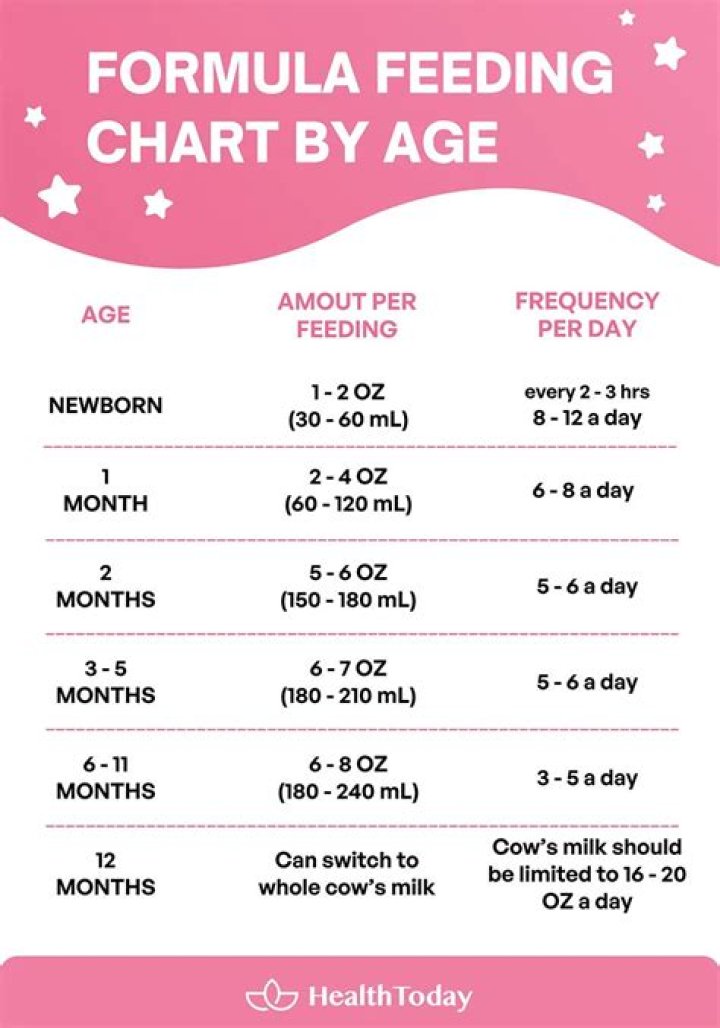
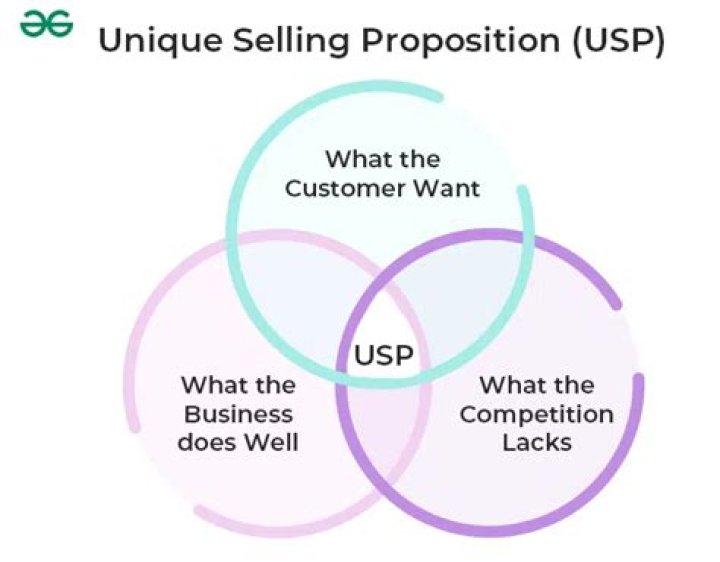
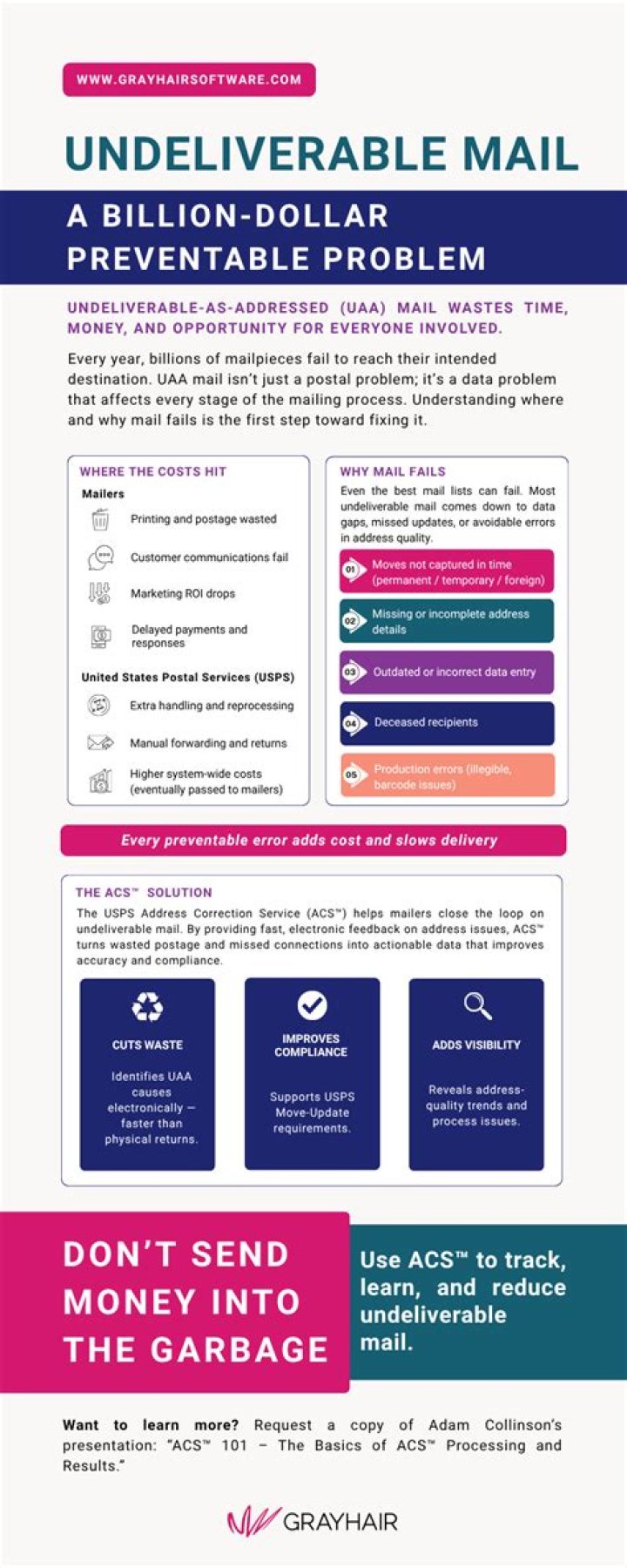
Can IR spectroscopy detect metals?
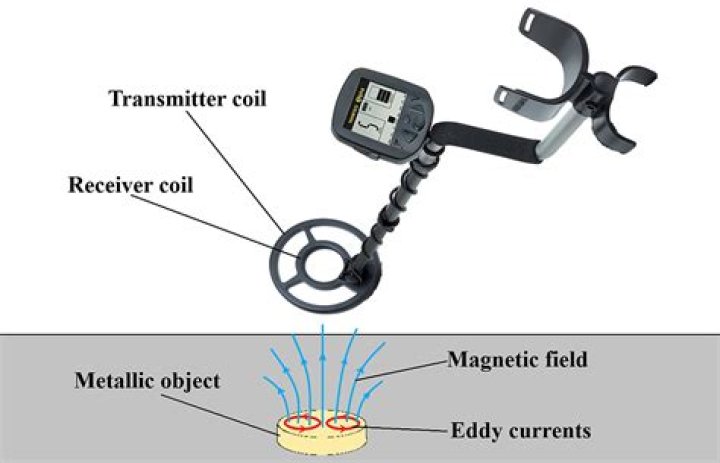
Can IR spectroscopy detect metals?
Infrared spectroscopy has proven to have potential in the analysis and concentration of heavy metal contamination [2,6,8]. Application of the FT-IR techniques for the investigation of heavy metal elements has been significantly developed due to their flexibility and accuracy [7,8,13,14].
Which of the molecules will show IR spectrum?
1. Homodiatomic molecules such as H2 are IR inactive. HCl and H2 O have a dipole moment. So, they are IR active.
What functional group does cyclohexanol have?
The presence of a hydroxyl group makes this molecule an alcohol (thus “hexanol”). The longest carbon chain is a ring structure (thus “cyclohexanol”), and the location of the alcohol group is assumed to be carbon 1 because it’s the highest priority functional group on the molecule.
What is the structure of cyclohexanol?
C6H12O
Cyclohexanol/Formula
Does FTIR work on metals?
Based on our observation, FTIR technique proves to be an efficient tool for detecting structural changes and probable binding sites induced by the presence of a metal pollutant, cadmium, in the marine environment.
Is H2 rotationally active?
Due to the dipole requirement, molecules such as HF and HCl have pure rotational spectra and molecules such as H2 and N2 are rotationally inactive.
What does an IR spectrum show?
The IR spectra tell you what types of vibrational modes (motion) the molecule responds with after it absorbs that light, and when you figure out which peaks correspond to which motions, you can figure out what functional groups the molecule has and (almost) what the molecule is.
What kind of compound is cyclohexanol?
Cyclohexanol is an alcohol that consists of cyclohexane bearing a single hydroxy substituent. The parent of the class of cyclohexanols. It has a role as a solvent. It is a secondary alcohol and a member of cyclohexanols.
What is the density of cyclohexanol?
962 kg/m³
Cyclohexanol/Density
How do you explain IR spectra?
An IR spectrum can be visualized in a graph of infrared light absorbance (or transmittance) on the vertical axis vs. frequency or wavelength on the horizontal axis. Typical units of frequency used in IR spectra are reciprocal centimeters (sometimes called wave numbers), with the symbol cm−1.
Which of the region of IR spectra appears between?
Explanation: Vibrational spectroscopy involves the transitions falling in the spectral range of 400-4000 cm-1 (infrared region). 2. Which of the region of IR spectra appears between (1400-600) cm-1? 3.