If the molecule is nonpolar, then no dipole-dipole interactions or hydrogen bonding can occur and the only possible intermolecular force is the weak van der Waals force..
Subsequently, one may also ask, can nonpolar molecules form hydrogen bonds?
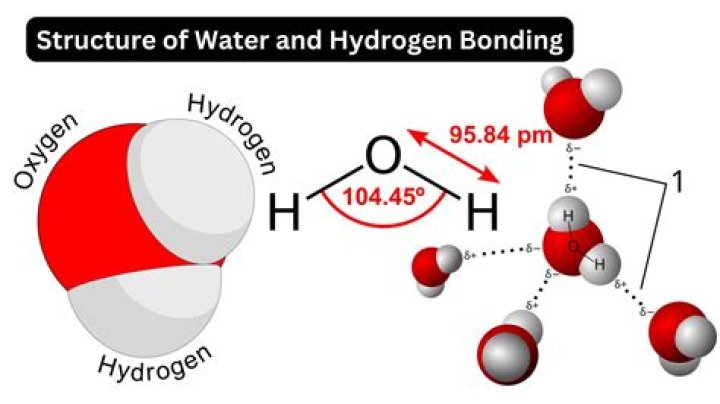
Water Is Polar The hydrogen and oxygen atoms within water molecules form polar covalent bonds. The shared electrons spend more time associated with the oxygen atom than they do with hydrogen atoms. Hydrogen bonds are not readily formed with nonpolar substances like oils and fats (Figure 1).
Subsequently, question is, what types of molecules will display hydrogen bonding? Hydrogen bond is formed only by the three highly electronegative elements- fluorine, oxygen and nitrogen. So, hydrogen bonding is possible only in those compounds in which the hydrogen atom is directly bonded to fluorine, oxygen or nitrogen.
Also to know is, do hydrogen bonds only occur polar molecules?
The hydrogen bond in polar molecules occurs only in compounds that have hydrogen bonded to N, O, or F. The H atom is attracted to the partial negative charge on an N, O, or F atom in another molecule. The hydrogen bond is an attraction but not a true chemical bond such as ionic or covalent bonds.
What type of bond is water?
Water is a polar molecule A water molecule is formed when two atoms of hydrogen bond covalently with an atom of oxygen. In a covalent bond electrons are shared between atoms. In water the sharing is not equal. The oxygen atom attracts the electrons more strongly than the hydrogen.
Related Question Answers
Is water polar or nonpolar?
Water (H2O) is polar because of the bent shape of the molecule. The reason the shape of the molecule isn't linear and nonpolar (e.g., like CO2) is because of the difference in electronegativity between hydrogen and oxygen. The electronegativity value of hydrogen is 2.1, while the electronegativity of oxygen is 3.5.How are hydrogen bonds formed?
A hydrogen bond is formed when the positive end of one molecule is attracted to the negative end of another. The concept is similar to magnetic attraction where opposite poles attract. Hydrogen has one proton and one electron. This makes hydrogen an electrically positive atom because it has a deficiency of electrons.Is ethanol polar or nonpolar?
Ethanol is a very polar molecule due to its hydroxyl (OH) group, with the high electronegativity of oxygen allowing hydrogen bonding to take place with other molecules. Ethanol therefore attracts polar and ionic molecules. The ethyl (C2H5) group in ethanol is non-polar. Ethanol therefore attracts non-polar molecules.Is Sugar polar or nonpolar?
Table sugar (sucrose) is a polar nonelectrolyte. Sucrose is quite soluble because its molecules bristle with water-accessible OH groups, which can form strong hydrogen bonds with water. So sugar is not an exception to the "like dissolves like" rule of thumb. Nonelectrolytes can be either polar or nonpolar.Why are hydrogen bonds important?
Hydrogen bonding is important in many chemical processes. Hydrogen bonding is responsible for water's unique solvent capabilities. Hydrogen bonds hold complementary strands of DNA together, and they are responsible for determining the three-dimensional structure of folded proteins including enzymes and antibodies.What is polar and nonpolar covalent bonds?
Polar covalent bonding is a type of chemical bond where a pair of electrons is unequally shared between two atoms. If the electronegativity of two atoms is basically the same, a nonpolar covalent bond will form, and if the electronegativity is slightly different, a polar covalent bond will form.What happens when you mix polar and nonpolar substances?
Polarity and Mixing Solutions The general rule is that "like dissolves like", which means polar molecules will dissolve into other polar liquids and nonpolar molecules will dissolve into nonpolar liquids. This is why oil and water don't mix: oil is nonpolar while water is polar.Is a hydrogen bond polar or nonpolar?
a hydrogen bond is formed when a charged part of a molecule having polar covalent bonds forms an electrostatic (charge, as in positive attracted to negative) interaction with a substance of opposite charge. Molecules that have nonpolar covalent bonds donot form hydrogen bonds.Are all hydrogen bonds polar?
Hydrogen bonds are intermolecular forces rather than forces within a molecule. These forces result from a bond where hydrogen in bound to an electronegative molecule resulting in a dipole. Therefore the intermolecular bond (within the molecule) between the electronegative atom and the hydrogen is polar.Are hydrogen bonds permanent?
In the liquid state, the hydrogen bonds of water can break and reform as the molecules flow from one place to another. When water is cooled, the molecules begin to slow down. Eventually, when water is frozen to ice, the hydrogen bonds become permanent and form a very specific network.Is ch3oh a hydrogen bond?
Methanol exhibits hydrogen bonding, and all three of the van der Waals attractions: Keesom forces (dipole-dipole) attraction, Debye forces (induced attraction) and London dispersion forces (which all molecules exhibit).Can hydrogen bonds be easily broken?
Hydrogen bonds This interaction is called a hydrogen bond. Hydrogen bonds are common, and water molecules in particular form lots of them. Individual hydrogen bonds are weak and easily broken, but many hydrogen bonds together can be very strong.What type of bond is a hydrogen bond?
A hydrogen bond is the electromagnetic attraction between polar molecules in which hydrogen is bound to a larger atom, such as oxygen or nitrogen. This is not a sharing of electrons, as in a covalent bond. Instead, this is an attraction between the positive and negative poles of charged atoms.Why is water a polar molecule?
A water molecule, because of its shape, is a polar molecule. That is, it has one side that is positively charged and one side that is negatively charged. The molecule is made up of two hydrogen atoms and one oxygen atom. The bonds between the atoms are called covalent bonds, because the atoms share electrons.Is a hydrogen bond equivalent to a covalent bond?
Hydrogen bond is the name given to the electrostatic interaction between the positive charge on a hydrogen atom and the negative charge on the oxygen atom of a neighboring molecule. The covalent bond is the electrostatic interaction between two atoms in the same molecule.Is water a hydrogen bond?
Hydrogen-bonding forms in liquid water as the hydrogen atoms of one water molecule are attracted towards the oxygen atom of a neighboring water molecule; generally, a proton shared by two lone electron pairs. This attraction is the basis of the 'hydrogen' bonds.Is ch4 a hydrogen bond?
Answer and Explanation: CH4 cannot form hydrogen bonds. This is because hydrogen bonds are a type of electrostatic interaction, which is only possible in molecules in whichIs ethanol a hydrogen bond?
Hydrogen bonding in alcohols Any molecule which has a hydrogen atom attached directly to an oxygen or a nitrogen is capable of hydrogen bonding. However, ethanol has a hydrogen atom attached directly to an oxygen - and that oxygen still has exactly the same two lone pairs as in a water molecule.When can hydrogen bonding occur?
A hydrogen bond is a weak type of force that forms a special type of dipole-dipole attraction which occurs when a hydrogen atom bonded to a strongly electronegative atom exists in the vicinity of another electronegative atom with a lone pair of electrons.