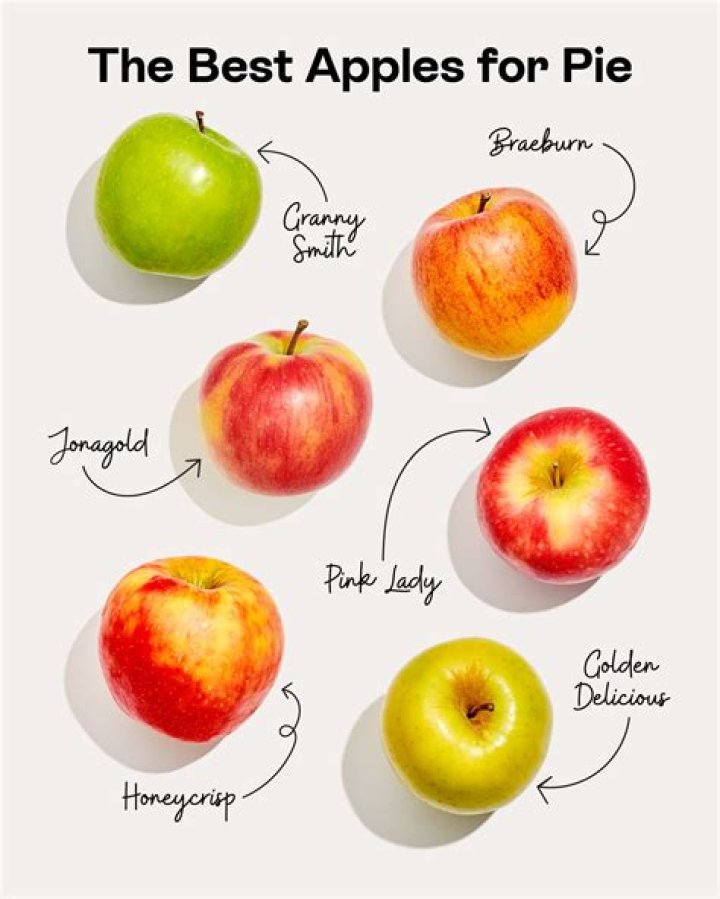
Are there any exceptions to the Pauli exclusion principle?
Are there any exceptions to the Pauli exclusion principle?
Particles with an integer spin, or bosons, are not subject to the Pauli exclusion principle: any number of identical bosons can occupy the same quantum state, as with, for instance, photons produced by a laser or atoms in a Bose–Einstein condensate.
What is a violation of the Pauli exclusion principle?
The Pauli Exclusion Principle states that, in an atom or molecule, no two electrons can have the same four electronic quantum numbers. Otherwise they will have the same four quantum numbers, in violation of the Pauli Exclusion Principle.
What are the 3 primary purposes of the exclusionary rule?
The prohibition of “using” such illegally obtained evidence applies to: (1) all stages of criminal proceedings; (2) direct and derivative evidence; (3) evidence obtained by private actors and evidence obtained by state officials; and (4) cases in which a third party’s rights, not the defendant’s, were violated.
What is prohibited under the exclusionary rule?
The exclusionary rule prevents the government from using most evidence gathered in violation of the United States Constitution. The decision in Mapp v. Ohio established that the exclusionary rule applies to evidence gained from an unreasonable search or seizure in violation of the Fourth Amendment.
What elements have exceptions to the electron configuration?
There are two main exceptions to electron configuration: chromium and copper.
Why two electrons Cannot be accommodated with the parallel spin in the same orbital?
Pauli’s Exclusion Principle states that no two electrons in the same atom can have identical values for all four of their quantum numbers. In other words, (1) no more than two electrons can occupy the same orbital and (2) two electrons in the same orbital must have opposite spins (Figure 46(i) and (ii)).
Which elements are exceptions to the Aufbau principle?
For example, ruthenium, rhodium, silver and platinum are all exceptions to the Aufbau principle because of filled or half-filled subshells. In the lower atomic numbers, the difference in energy levels for the normal sequence of electron shells is larger and exceptions are not as common.
Why 1S3 is not possible?
The 1S3 configuration is forbidden by a fundamental feature of nature summarized by the Austrian physicist Wolfgang Pauli in the Pauli exclusion principle. After filling 2 electrons in 1S orbital, remaining electrons are assigned to the next coming orbitals…